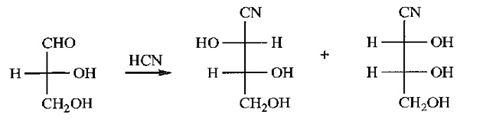
Chapter 23: Q50P (page 1255)
(a) Give the products expected when (-)-erythrose reacts with HCN.
(b) What is the relationship between the products? How might they be separated?
(c) Are the products optically active? Explain.
Short Answer
(a) (-)-erythrose:
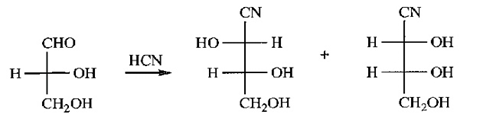
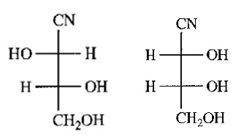
(b) The relationship between the products is that they are diastereomers of each other. They could be separated by crystallization, distillation process.
(c) Yes, both the products are optically active.