Chapter 23: Q47P (page 1255)
Without referring to the chapter, draw the chair conformation of
(a) α -D-mannopyranose (the C2 epimer of glucose).
(b) β -D-allopyranose (the C3 epimer of glucose.)
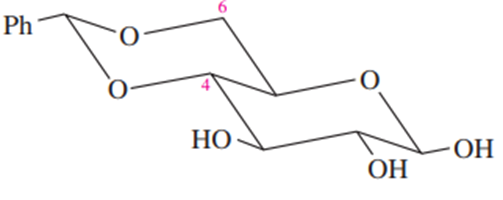
(c) α -D-galactopyranose (the C4 epimer of glucose).
(d) N-formylglucosamine, glucose with C2 oxygen atom replaced by a formylated amino group.
Short Answer
(a) α -D-mannopyranose (the C2 epimer of glucose).
(b) β -D-allopyranose (the C3 epimer of glucose.)
(c) α -D-galactopyranose (the C4 epimer of glucose).
(d) N-formylglucosamine, glucose with C2 oxygen atom replaced by a formylated amino group.