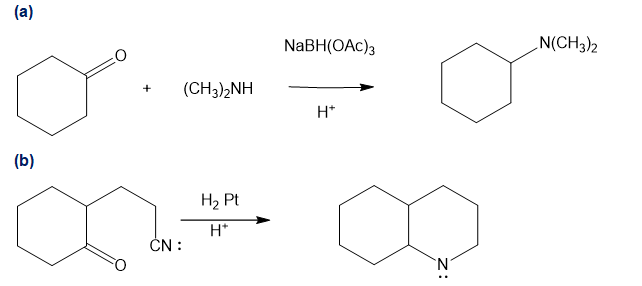
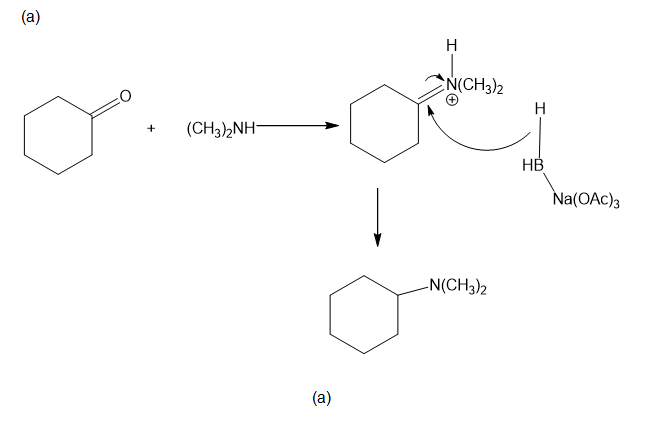
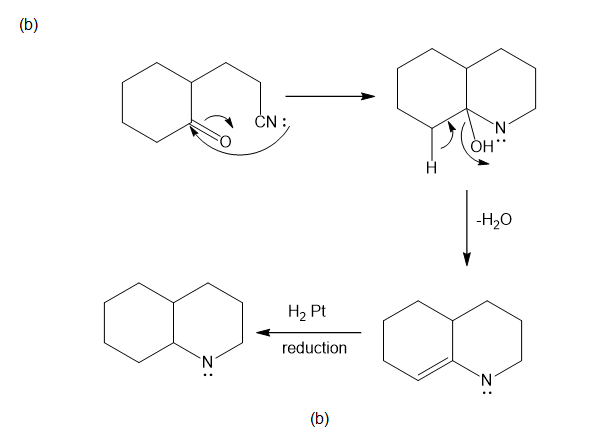
Chapter 19: Q46P (page 1031)
Propose mechanisms for the following reactions.
Short Answer
Learning Materials
Features
Discover
Chapter 19: Q46P (page 1031)
Propose mechanisms for the following reactions.



All the tools & learning materials you need for study success - in one app.
Get started for free
Show how you would convert aniline to the following compounds
(a)flurobenzene (b) chlorobenzene
(c) 1,3,5- trimethylbenzene(d) bromobenzene
(e) iodobenzene(f) benzonitrile
(g) phenol
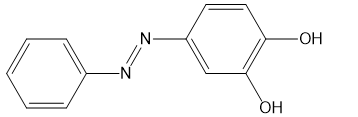
(h)
Predict the products from the reactions of the following amines with sodium nitrite in dilute.
The proton and 13C NMR spectra of a compound of formula C4H11N are shown here. Determine the structure of this amine, and give peak assignments for all of the protons in the structure.
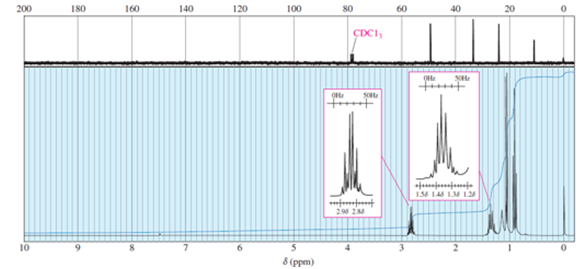
Question. The carbon NMR chemical shifts of diethylmethylamine, piperidine, propan-1-ol, and propanal follow. Determine which spectrum corresponds to each structure, and show which carbon atom(s) are responsible for each absorption.
(a) 25.9, 27.8, 47.9
(b) 12.4, 41.0, 51.1
(c) 7.9, 44.7, 201.9
(d) 10.0, 25.8, 63.6
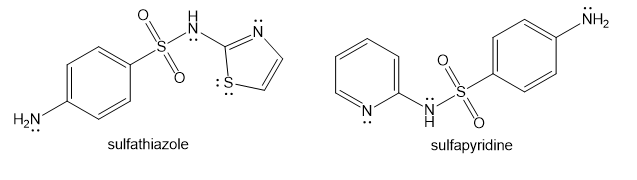
Show how you would use the same sulfonyl chloride as used in the sulfanilamide synthesis to make sulfathiazole and sulfapyridine.
What do you think about this solution?
We value your feedback to improve our textbook solutions.