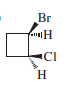
Chapter 6: Q50P (page 330)
Give a mechanism to explain the two products formed in the following reaction.
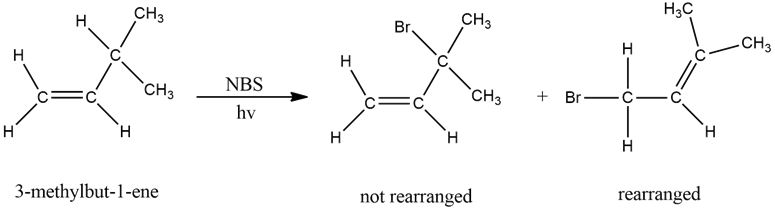
Short Answer
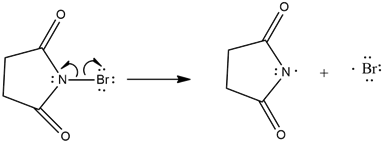
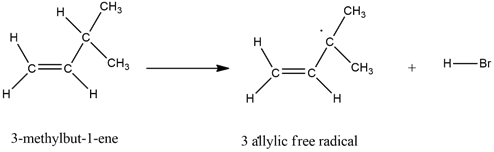
The mechanism of the given reaction is as follows,

Learning Materials
Features
Discover
Chapter 6: Q50P (page 330)
Give a mechanism to explain the two products formed in the following reaction.

The mechanism of the given reaction is as follows,



All the tools & learning materials you need for study success - in one app.
Get started for free
Question: Using cyclohexane as one of your starting materials, show how you would synthesize the following compounds.
d)
For each pair of compounds, predict which one has the higher molecular dipole moment, and explain your reasoning.
(a) ethyl chloride or ethyl iodide
(b) 1-bromopropane or cyclopropane
(c) cis-2,3-dibromobut-2-ene or trans-2,3-dibromobut-2-ene
(d) cis-1,2-dichlorocyclobutane or trans-1,3-dichlorocyclobutane.
Question: For each of the following compounds,
a)
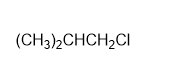
b)
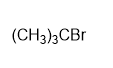
c)
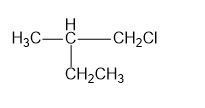
d)
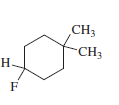
e)
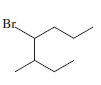
f)
Strawberry growers have used large quantities of methyl bromide (b.p. 4°C) to sterilize the soil before planting their crops. Like some of the freons, methyl bromide can diffuse up into the stratosphere, where it damages the protective ozone layer. Agricultural chemists have suggested using methyl iodide (b.p. 43°C) as a replacement for methyl bromide. Why is methyl iodide likely to be more toxic to agricultural pests (and people) than methyl bromide? Why is methyl iodide less likely to reach the stratosphere than methyl bromide?
Kepone, aldrin, and chlordane are synthesized from hexachlorocyclopentadiene and other five-membered ring compounds. Show how these three pesticides are composed of two five-membered rings.

What do you think about this solution?
We value your feedback to improve our textbook solutions.