Chapter 6: 6-26P (page 320)
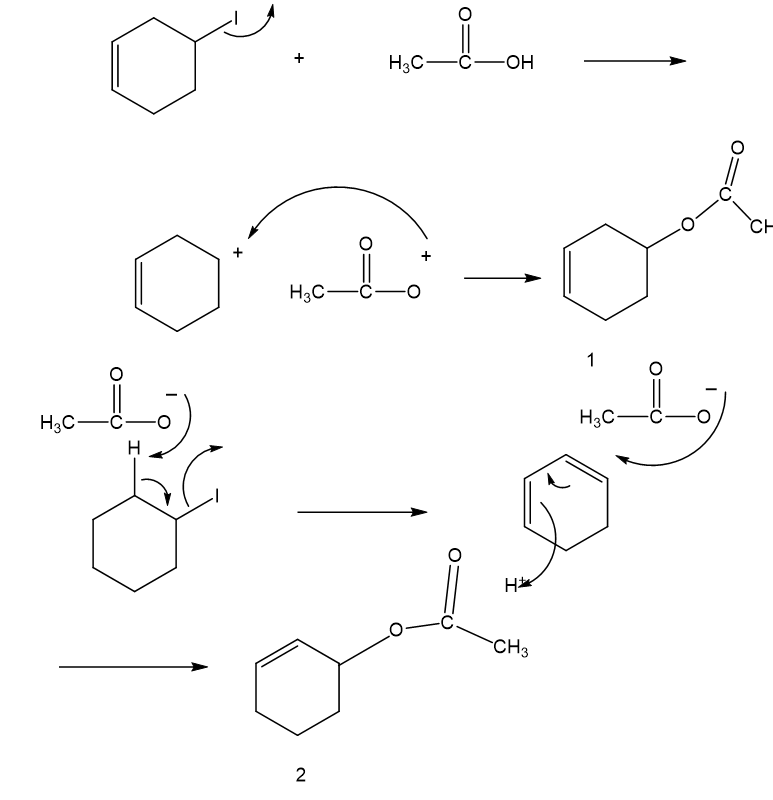
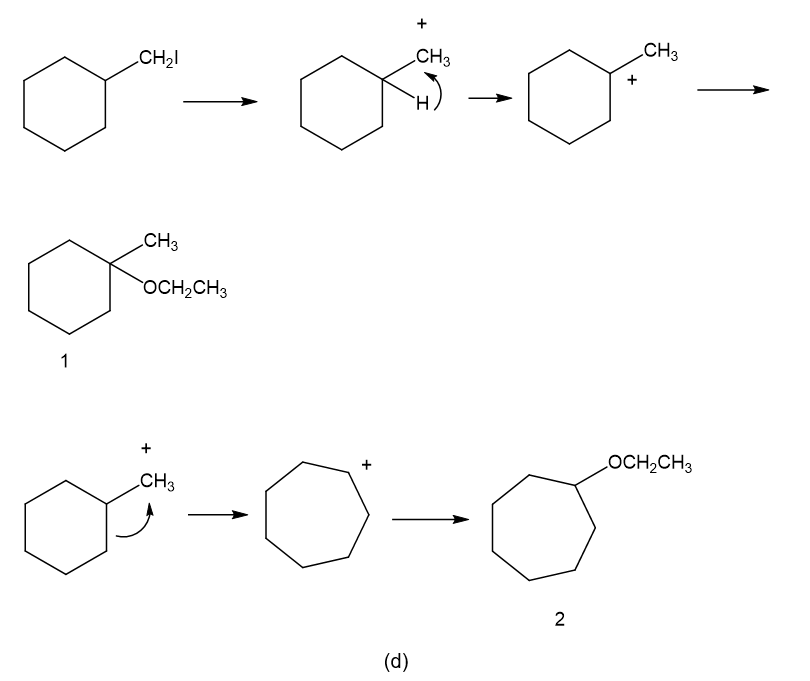
Propose a mechanism involving a hydride shift or alkyl shift for each solvolysis reaction. Explain howeach rearrangement forms a more stable intermediate.
(a)
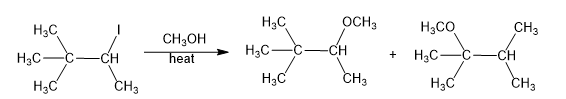
(b)
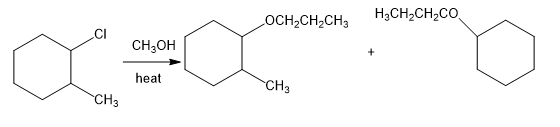
(c)
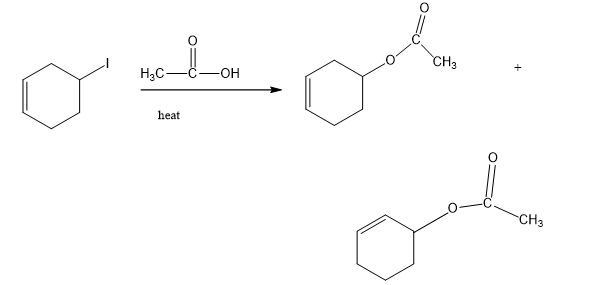
(d)
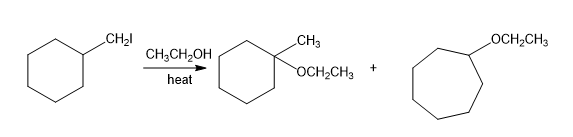
Short Answer
(a)
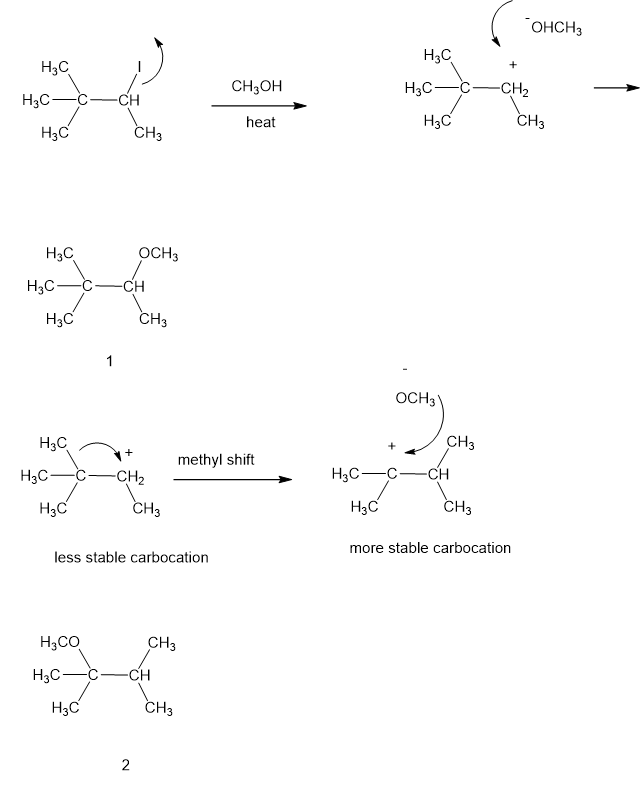
(b)

(c)
(d)
Learning Materials
Features
Discover
Chapter 6: 6-26P (page 320)
Propose a mechanism involving a hydride shift or alkyl shift for each solvolysis reaction. Explain howeach rearrangement forms a more stable intermediate.
(a)

(b)

(c)

(d)

(a)

(b)

(c)

(d)

All the tools & learning materials you need for study success - in one app.
Get started for free
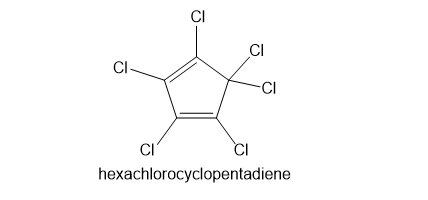
Kepone, aldrin, and chlordane are synthesized from hexachlorocyclopentadiene and other five-membered ring compounds. Show how these three pesticides are composed of two five-membered rings.
Question: Using cyclohexane as one of your starting materials, show how you would synthesize the following compounds.
e)
Predict the compound in each pair that will undergo the SN2 reaction faster.
(a.)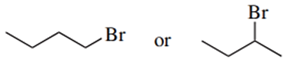
(b.)
(c.)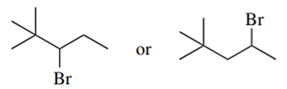
(d.)
(e.)
(f.)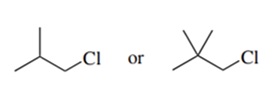
Question: Using cyclohexane as one of your starting materials, show how you would synthesize the following compounds.
d)
Classify each compound as an alkyl halide, a vinyl halide, or an aryl halide.
(a) (CH3)3CF
(b) CH3CHCBrCH3
(c) CH3CF3
(d)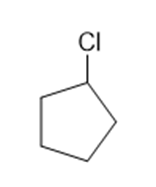
(e)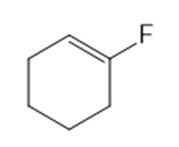
(f)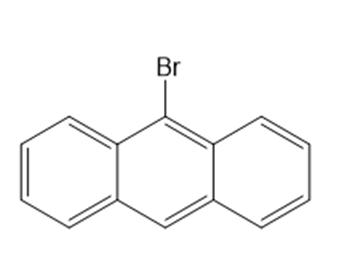
What do you think about this solution?
We value your feedback to improve our textbook solutions.