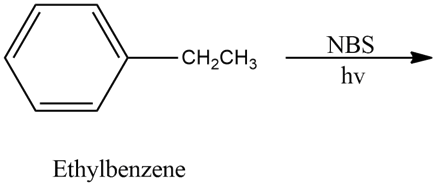
Chapter 6: 58 P-d (page 331)
Question: Show the products you expect when each compound reacts with NBS with light shining on the reaction.
d)
Short Answer
d)The product formed from the above compound when react with NBS
Learning Materials
Features
Discover
Chapter 6: 58 P-d (page 331)
Question: Show the products you expect when each compound reacts with NBS with light shining on the reaction.
d)
d)The product formed from the above compound when react with NBS
All the tools & learning materials you need for study success - in one app.
Get started for free
Give the structures of the following compounds.
(a) Methylene chloride
(b) Carbon tetrachloride
(c) 3-iodo-2-methylpentane
(d) Chloroform
(e) 2-chloro-3-ethyl-2-methyhexane
(f) Isobutyl iodide
(g) Cis-1-chloro-3-(chloromethyl)cyclohexane
(h) Tert-butyl bromide
(a)Optically active 2-bromobutane undergoes racemization on treatment with a solution of KBr. Give a mechanism for this racemization.
(b)In contrast, optically active butan-2-ol does not racemize on treatment with a solution of KOH. Explain why a reaction like that in part (a) does not occur.
(c)Optically active butan-2-ol racemizes in dilute acid. Propose a mechanism for this racemization.
(c)Optically active butan-2-ol racemizes in dilute acid. Propose a mechanism for this racemization.
Predict the major product of the following reaction, and give a mechanism to support your prediction.
Question: Propose mechanisms to account for the observed products in the following reactions.
b)
What do you think about this solution?
We value your feedback to improve our textbook solutions.