Chapter 6: 49 P-c (page 330)
(c)Optically active butan-2-ol racemizes in dilute acid. Propose a mechanism for this racemization.
Short Answer
c)
Learning Materials
Features
Discover
Chapter 6: 49 P-c (page 330)
(c)Optically active butan-2-ol racemizes in dilute acid. Propose a mechanism for this racemization.
c)
All the tools & learning materials you need for study success - in one app.
Get started for free
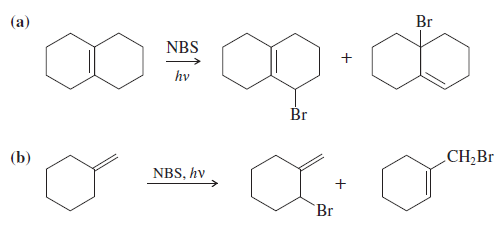
Question: Propose mechanisms to account for the observed products in the following reactions.
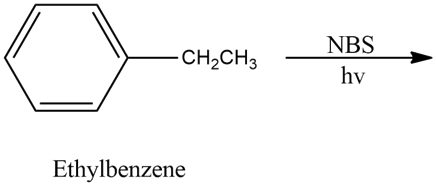
Predict the major product of the following reaction, and give a mechanism to support your prediction.
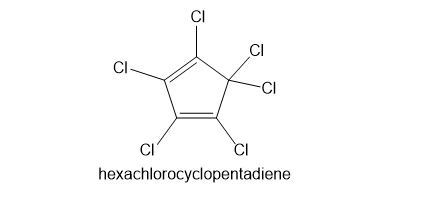
Kepone, aldrin, and chlordane are synthesized from hexachlorocyclopentadiene and other five-membered ring compounds. Show how these three pesticides are composed of two five-membered rings.
For each of the following compounds,
(b)In contrast, optically active butan-2-ol does not racemize on treatment with a solution of KOH. Explain why a reaction like that in part (a) does not occur.
What do you think about this solution?
We value your feedback to improve our textbook solutions.