(a)
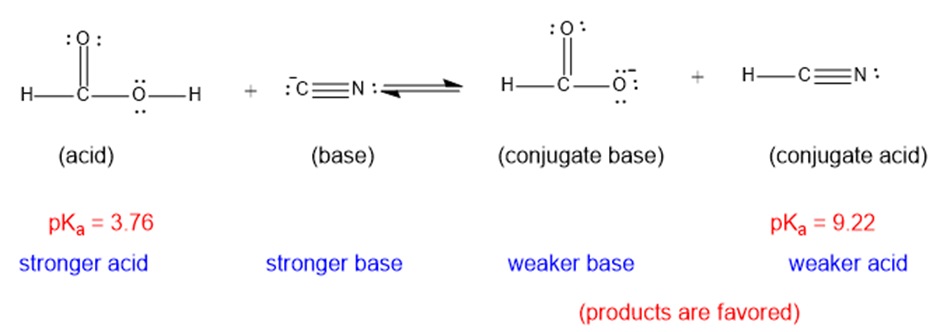
The weaker acid and weaker base are both present in the product side, hence the equilibrium will favor the products.
(b)
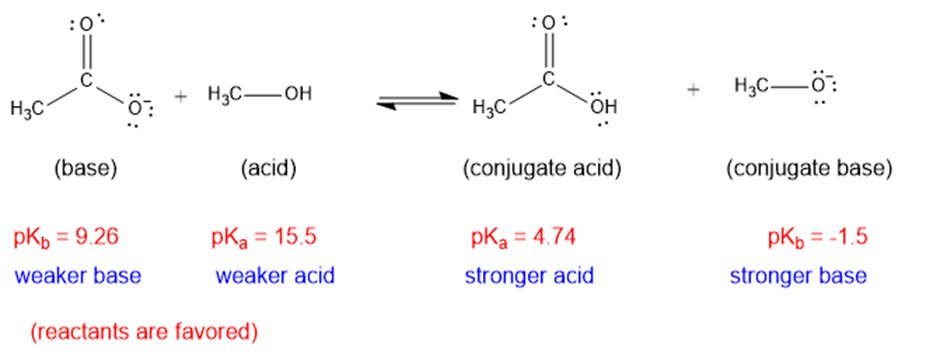
The weaker acid and weaker base are both present in the reactant side, hence the equilibrium will favor the reactants.
(c)
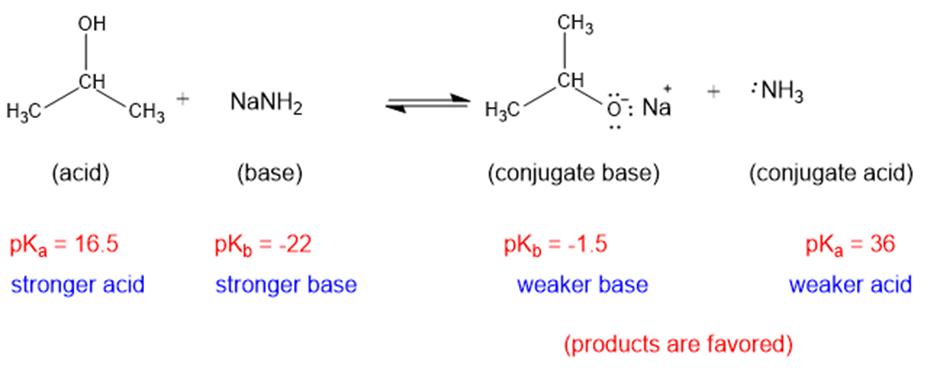
The weaker acid and weaker base are both present in the product side, hence the equilibrium will favor the products.
(d)
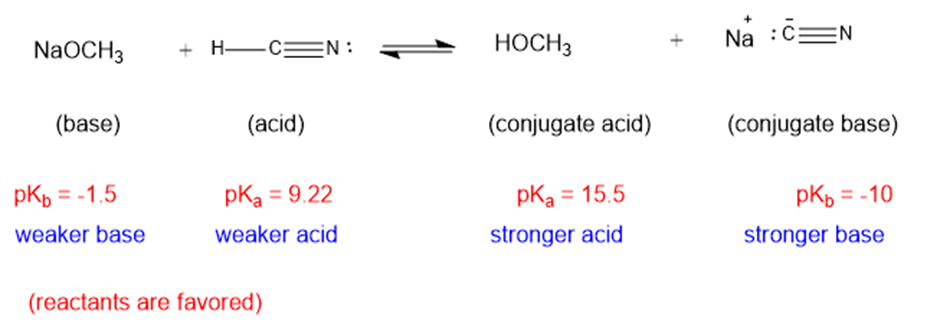
The weaker acid and weaker base are both present in the reactant side, hence the equilibrium will favor the reactants.
(e)
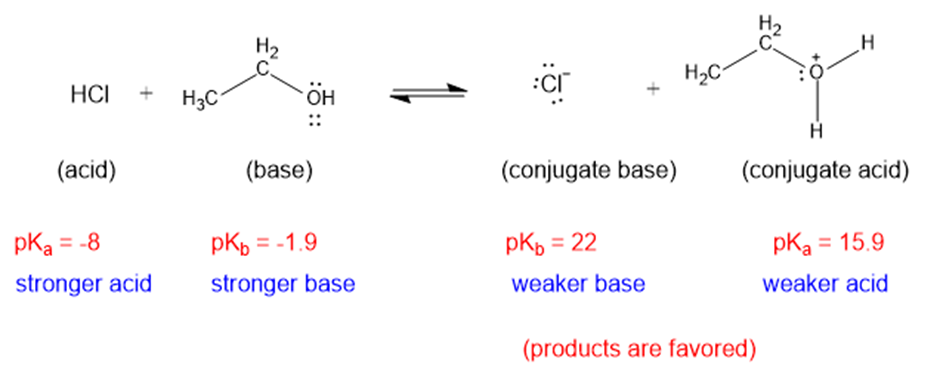
The weaker acid and weaker base are both present in the product side, hence the equilibrium will favor the products.
(f)
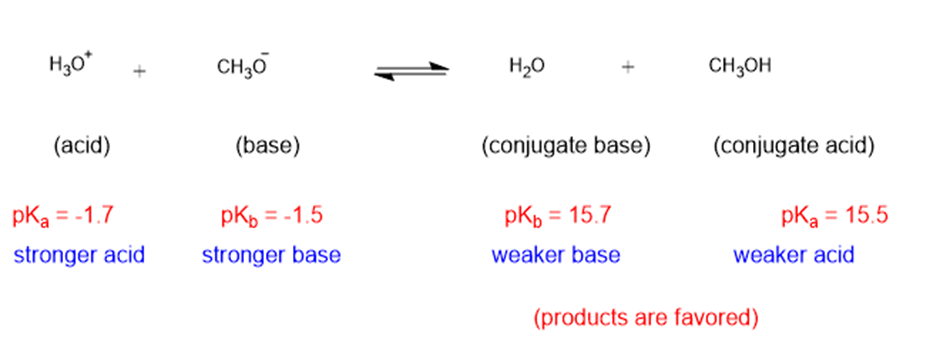
The weaker acid and weaker base are both present in the product side, hence the equilibrium will favor the products.
(g)
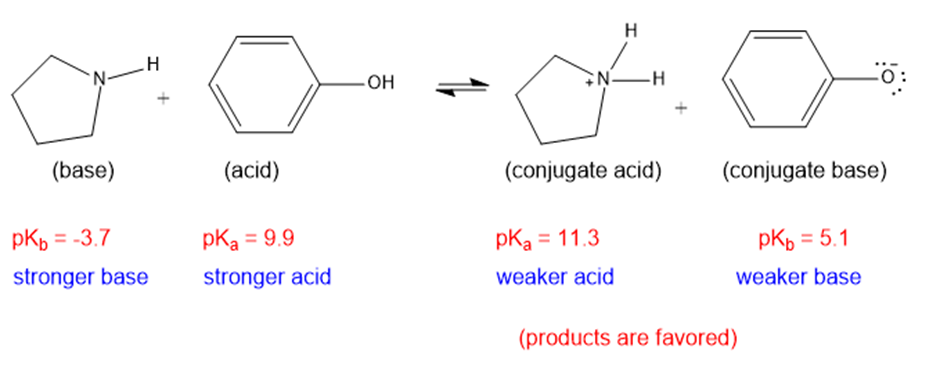
The weaker acid and weaker base are both present in the product side, hence the equilibrium will favor the products.
(h)
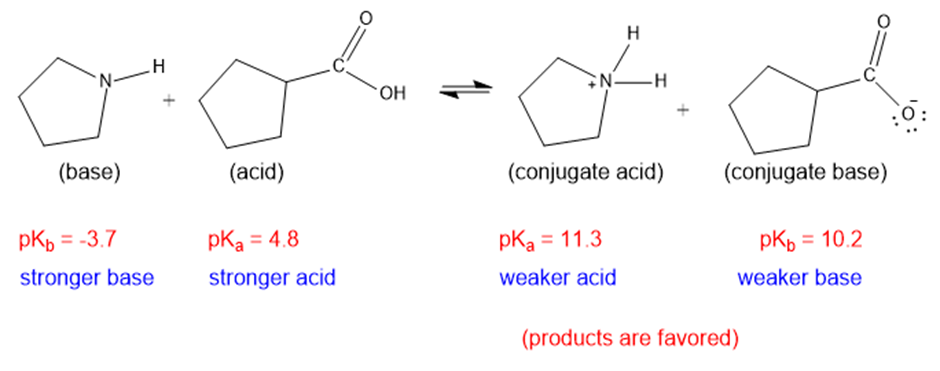
The weaker acid and weaker base are both present in the product side, hence the equilibrium will favor the products.
(i)
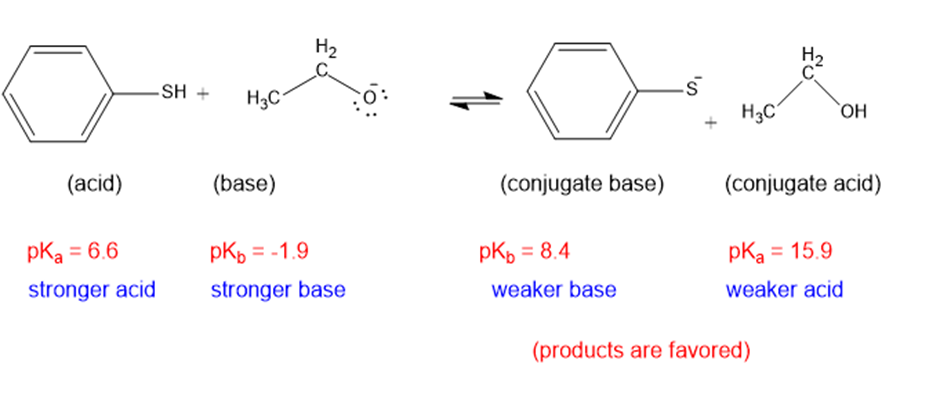
The weaker acid and weaker base are both present in the product side, hence the equilibrium will favor the products.
(j)
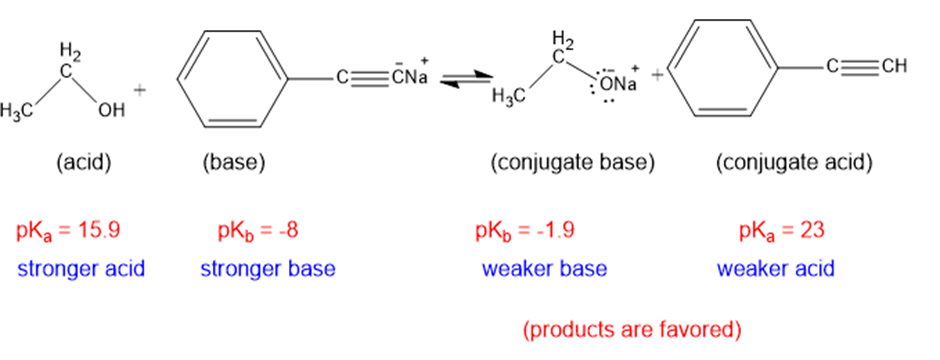
The weaker acid and weaker base are both present in the product side, hence the equilibrium will favor the products.







































