Chapter 27: Q 24. (page 1096)
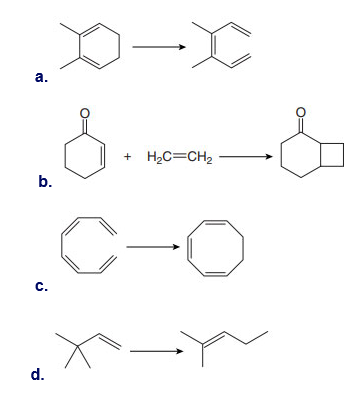
Question: What product is formed from the Claisen rearrangement of each starting material?
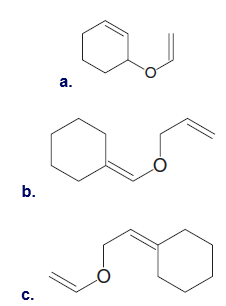
Short Answer
Answer
a. 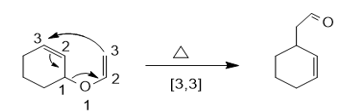
b. 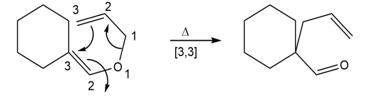
c. 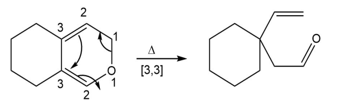
Learning Materials
Features
Discover
Chapter 27: Q 24. (page 1096)
Question: What product is formed from the Claisen rearrangement of each starting material?

Answer
a. 
b. 
c. 
All the tools & learning materials you need for study success - in one app.
Get started for free
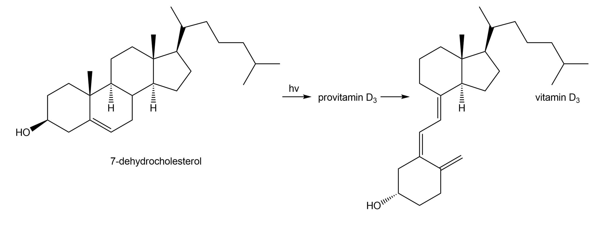
Question: Vitamin , the most abundant of the D vitamins, is synthesized from 7-dehydrocholesterol, a compound found in milk and fatty fish, such as salmon and mackerel. When the skin is exposed to sunlight, a photochemical electrocyclic ring-opening forms provitamin , which is then converted to vitamin by a sigmatropic rearrangement (Section 27.5). Draw the structure of provitamin .
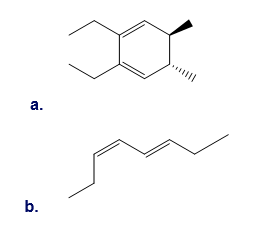
Question: What product is formed when each compound undergoes thermal electrocyclic ring opening or ring closure? Label each process as conrotatory or disrotatory and clearly indicate the stereochemistry around tetrahedral stereogenic centers and double bonds.
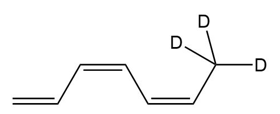
Question:(a) What product is formed from the [1,7] sigmatropic rearrangement of a deuterium in the following triene?
(b) Does this reaction proceed in a suprafacial or antarafacial manner under
thermal conditions?
(c) Does this reaction proceed in a suprafacial or antarafacial manner under
photochemical conditions?
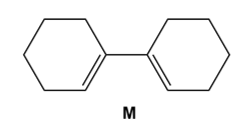
Question: Draw the product formed when diene M undergoes disrotatory cyclization. Indicate the stereochemistry at new hybridized carbons. Will the reaction occur under thermal or photochemical conditions?
Question: Classify each reaction as an electrocyclic reaction, a cycloaddition, or a sigmatropic rearrangement. Label the σ bonds that are broken or formed in each reaction.
What do you think about this solution?
We value your feedback to improve our textbook solutions.