Chapter 27: Q 12. (page 1088)
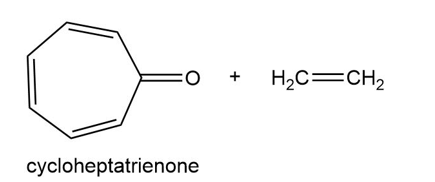
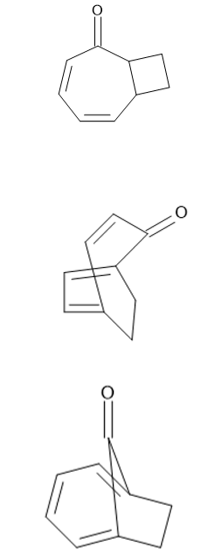
Question:Consider cycloheptatrienone and ethylene, and draw a possible product formed from each type of cycloaddition:
(a) [2 + 2]
(b) [4 + 2]
(c) [6 + 2]
Short Answer
Answer
Learning Materials
Features
Discover
Chapter 27: Q 12. (page 1088)
Question:Consider cycloheptatrienone and ethylene, and draw a possible product formed from each type of cycloaddition:
(a) [2 + 2]
(b) [4 + 2]
(c) [6 + 2]

Answer

All the tools & learning materials you need for study success - in one app.
Get started for free
Question: Draw the product of each electrocyclic reaction.
a. the thermal electrocyclic ring closure of (2E,4Z,6Z)-nona-2,4,6-triene
b. the photochemical electrocyclic ring closure of (2E,4Z,6Z)-nona-2,4,6-triene
c. the thermal electrocyclic ring-opening of cis-5-ethyl-6-methylcyclohexa-1,3-diene
d. the photochemical electrocyclic ring-opening of trans-5-ethyl-6-methylcyclohexa-1,3- diene
Question: (a) Using Figure 27.2 as a guide, draw the molecular orbitals for hexa-2,4-diene. (b) Label the HOMO and the LUMO in the ground state. (c) Label the HOMO and the LUMO in the excited state.
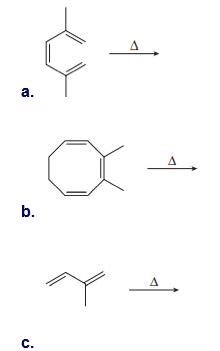
Question: Use curved arrows and draw the product of each electrocyclic reaction.
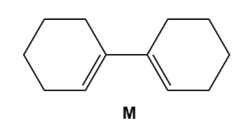
Question: Draw the product formed when diene M undergoes disrotatory cyclization. Indicate the stereochemistry at new hybridized carbons. Will the reaction occur under thermal or photochemical conditions?
Question: Classify each pericyclic reaction as an electrocyclic reaction, cycloaddition, or sigmatropic rearrangement. Indicate whether the stereochemistry is conrotatory, disrotatory, suprafacial, or antarafacial.
What do you think about this solution?
We value your feedback to improve our textbook solutions.