Chapter 27: Q 10. (page 1087)
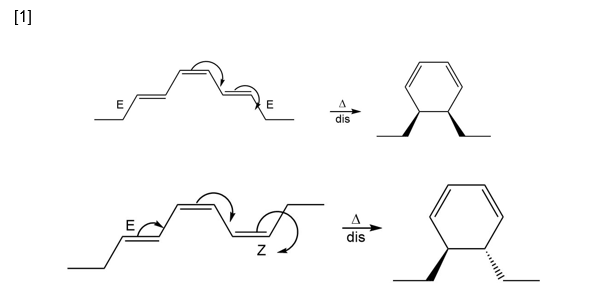
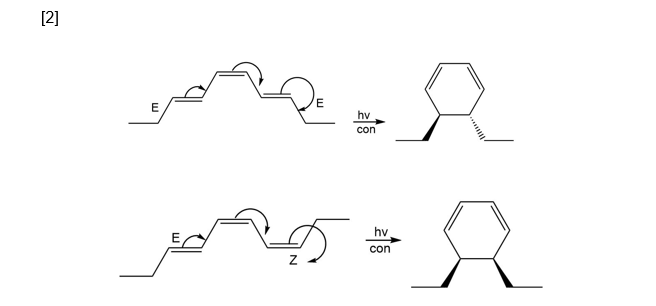
Question: Draw the product formed when each triene undergoes electrocyclic reaction under [1] thermal conditions, [2] photochemical conditions
Short Answer
Learning Materials
Features
Discover
Chapter 27: Q 10. (page 1087)
Question: Draw the product formed when each triene undergoes electrocyclic reaction under [1] thermal conditions, [2] photochemical conditions


All the tools & learning materials you need for study success - in one app.
Get started for free
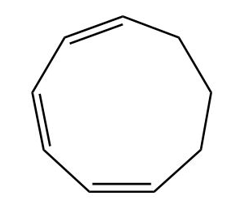
Question:What product would be formed by the disrotatory cyclization of the given triene? Would this reaction occur under photochemical or thermal conditions?
Question: For each molecular orbital in Figure 27.2, count the number of bonding interactions (interactions between adjacent orbitals of similar phase) and the number of nodes. (a) How do these two values compare for a bonding molecular orbital? (b) How do these two values compare for an antibonding molecular orbital?
Question: [4 + 2] Cycloadditions with o-quinones such as B are often complex because a variety of products are possible.a. Draw arrows to illustrate how each product is formed when B reacts with styrene, and label the “diene” and “dienophile” components.
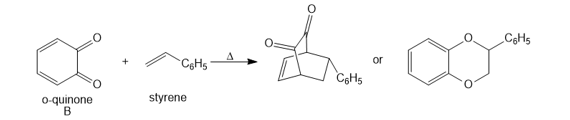
b. Draw arrows to illustrate how each product is formed when B reacts with buta-1,3-diene, and label the “diene” and “dienophile” components.
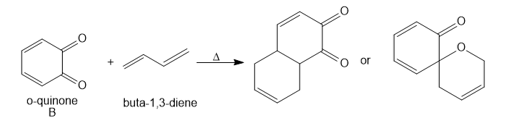
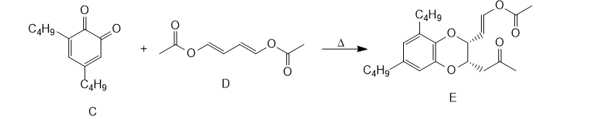
c. o-Quinone C reacts with diene D to form heterocycle E by a process that involves a cycloaddition followed by a [3,3] sigmatropic rearrangement. Use curved arrows to illustrate how this two-step sequence occurs. E is not formed directly from C by a Diels– Alder reaction.
Question: The bicyclic alkene P can be prepared by thermal electrocyclic ring closure from cyclodecadiene Q or by photochemical electrocyclic ring closure from cyclodecadiene R.Draw the structures of Q and R, and indicate the stereochemistry of the process by which each reaction occurs.
Question: Classify each pericyclic reaction as an electrocyclic reaction, cycloaddition, or sigmatropic rearrangement. Indicate whether the stereochemistry is conrotatory, disrotatory, suprafacial, or antarafacial.
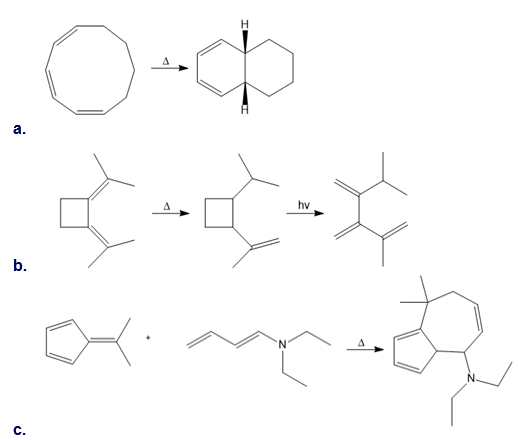
What do you think about this solution?
We value your feedback to improve our textbook solutions.