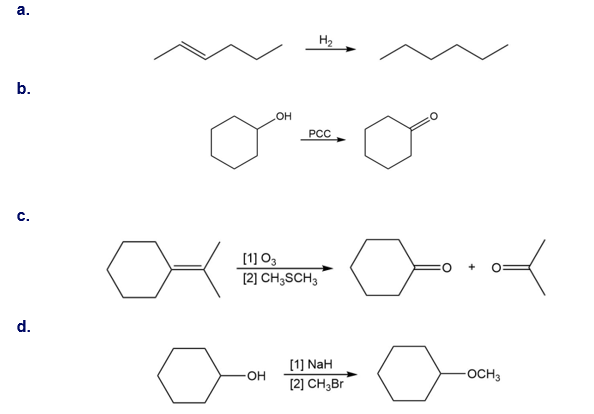
Chapter 13: Q 46. (page 523)
Question: Tell how IR spectroscopy could be used to determine when each reaction is complete.
Short Answer
Answer
The change in functional groups of the starting material of the product helps in predicting the product.
Learning Materials
Features
Discover
Chapter 13: Q 46. (page 523)
Question: Tell how IR spectroscopy could be used to determine when each reaction is complete.

Answer
The change in functional groups of the starting material of the product helps in predicting the product.
All the tools & learning materials you need for study success - in one app.
Get started for free
Question: The mass spectrum of the following compound shows fragments at m/z = 127, 113, and 85. Propose structures for the ions that give rise to these peaks.
Question:For each compound, assign likely structures to the fragments at each m/zvalue, and explain how each fragment is formed.
a. : peaks at m/z= 104, 91
b. : peaks at m/z= 71, 68, 41, 31
Question: Treatment of benzaldehyde () with Zn(Hg) in aqueous HCl forms a compound Z that has a molecular ion at 92 in its mass spectrum. Z shows absorptions at 3150–2950, 1605, and 1496 cm-1 in its IR spectrum. Give a possible structure for Z.
Question: Propose two molecular formulas for each molecular ion: (a) 102; (b) 98; (c) 119; (d) 74.
Question: The mass spectrum of 2,3-dimethylpentane also shows peaks at m/z = 57 and 43. Propose possible structures for the ions that give rise to these peaks.
What do you think about this solution?
We value your feedback to improve our textbook solutions.