Chapter 13: Mass Spectrometry and Infrared Spectroscopy
Q 37.
Question:Propose a structure consistent with each set of data.
a. a compound that contains a benzene ring and has a molecular ion at m/z= 107
b. a hydrocarbon that contains only sp3 hybridized carbons and a molecular ion at m/z= 84
c. a compound that contains a carbonyl group and gives a molecular ion at m/z= 114
d. a compound that contains C, H, N, and O and has an exact mass for the molecular ion at
101.0841
Q 38.
Question:A low-resolution mass spectrum of the neurotransmitter dopamine gave a molecular ion at m/z= 153. Two possible molecular formulas for this molecular ion are and . A high-resolution mass spectrum provided an exact mass at 153.0680. Which of the possible molecular formulas is the correct one?
Q 39.
Question:Primary (1°) alcohols often show a peak in their mass spectra at m/z= 31. Suggest a structure for this fragment.
Q 4.
Question: What molecular ions would you expect for compounds having each of the following molecular formulas: (a); (b); (c); (d)?
Q 40.
Question:Like alcohols, ethers undergo α cleavage by breaking a carbon–carbon bond between an alkyl group and the carbon bonded to the ether oxygen atom; that is, the red C–C bond in is broken. With this in mind, propose structures for the fragments formed by α cleavage of. Suggest a reason why an ether fragments by α cleavage.
Q 41.
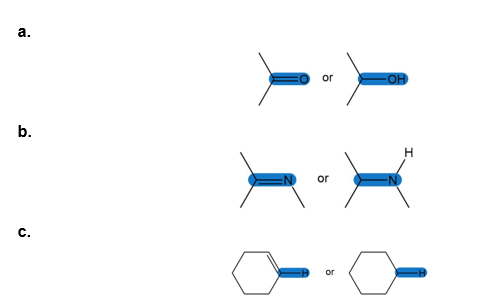
Question: Which of the highlighted bonds absorbs at higher in an IR spectrum?
Q 42.
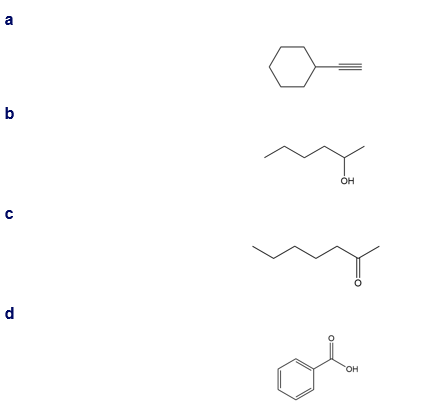
Question: What major IR absorptions are present above 1500 for each compound?
Q 43.
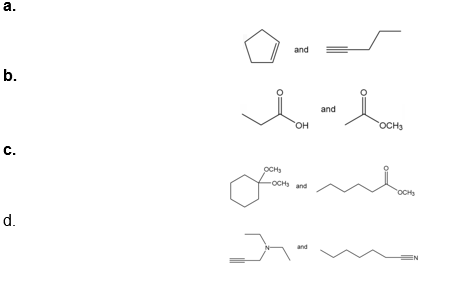
Question: How would each of the following pairs of compounds differ in their IR spectra?
Q 44.
Question: Morphine, heroin, and oxycodone are three addicting analgesic narcotics. How could IR spectroscopy be used to distinguish these three compounds from each other?
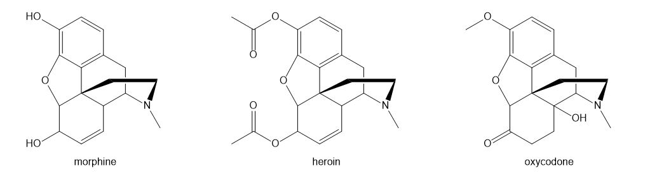
Q 45.
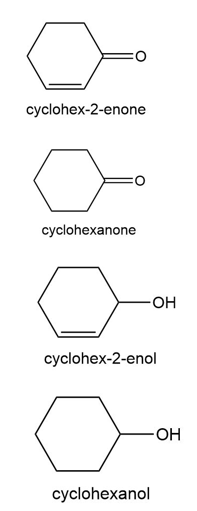
Question:Reduction of cyclohex-2-enone can yield cyclohexanone, cyclohex-2-enol, or cyclohexanol, depending on the reagent and reaction conditions. How could you use IR spectroscopy to distinguish the three possible products?