Chapter 25: Q51P (page 1032)
Question. Using any necessary reagents, show how you can accomplish the following multistep synthesis.
Learning Materials
Features
Discover
Chapter 25: Q51P (page 1032)
Question. Using any necessary reagents, show how you can accomplish the following multistep synthesis.
All the tools & learning materials you need for study success - in one app.
Get started for free
Question. Pyrrole undergoes electrophilic aromatic substitution more readily than benzene, and mild reagents and conditions are sufficient. These reactions normally occur at the 2-position rather than the 3-position, as shown in the following example.
 . Be careful to draw all the resonance structures of this intermediate.
. Be careful to draw all the resonance structures of this intermediate.Question. Which of the following compounds are capable of being resolved into enantiomers?
(a)N-ethyl-N-propylaniline
(b) 2-ethylpiperidine
(c) 1-ethylpiperidine
(d) 1,2,2-triethylaziridine
(e)
(f)
(g)
(h)
Question. Section 17-12 showed how nucleophilic aromatic substitution can give aryl amines if there is a strong electron-withdrawing group ortho or para to the site of substitution. Consider the following example.
(a) Propose a mechanism for this reaction.
(b) We usually think of fluoride ion as a poor leaving group. Explain why this reaction readily displaces fluoride as the leaving group.
(c) Explain why this reaction stops with the desired product, rather than reacting with another dinitrofluorobenzene.
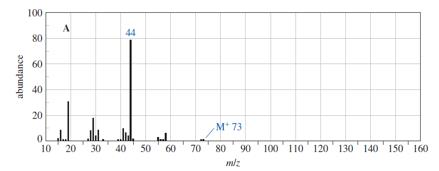
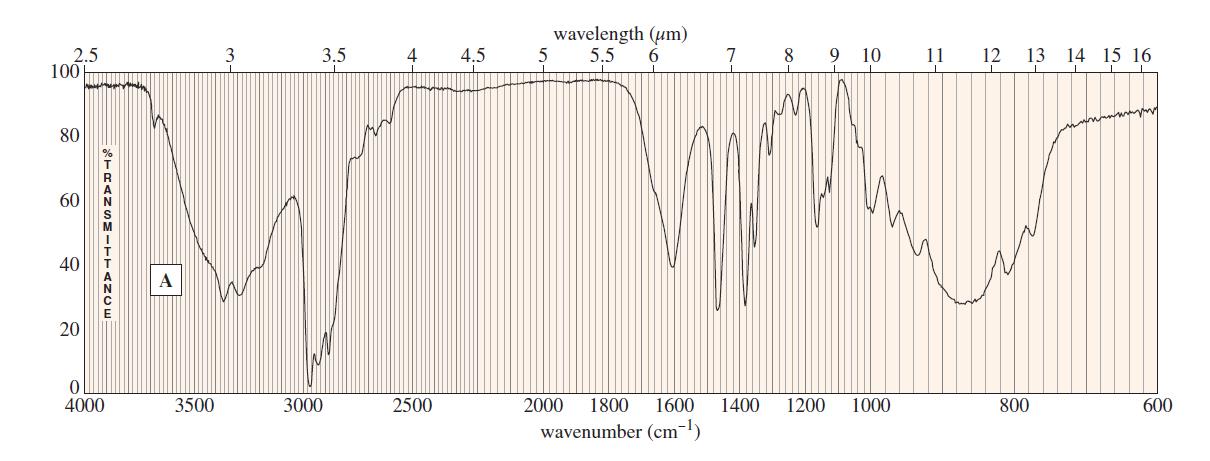
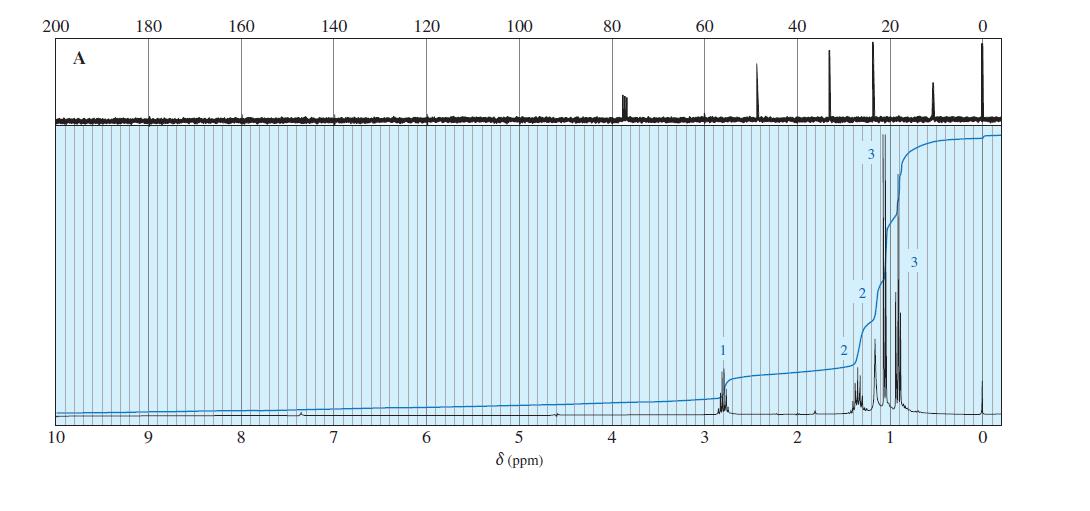
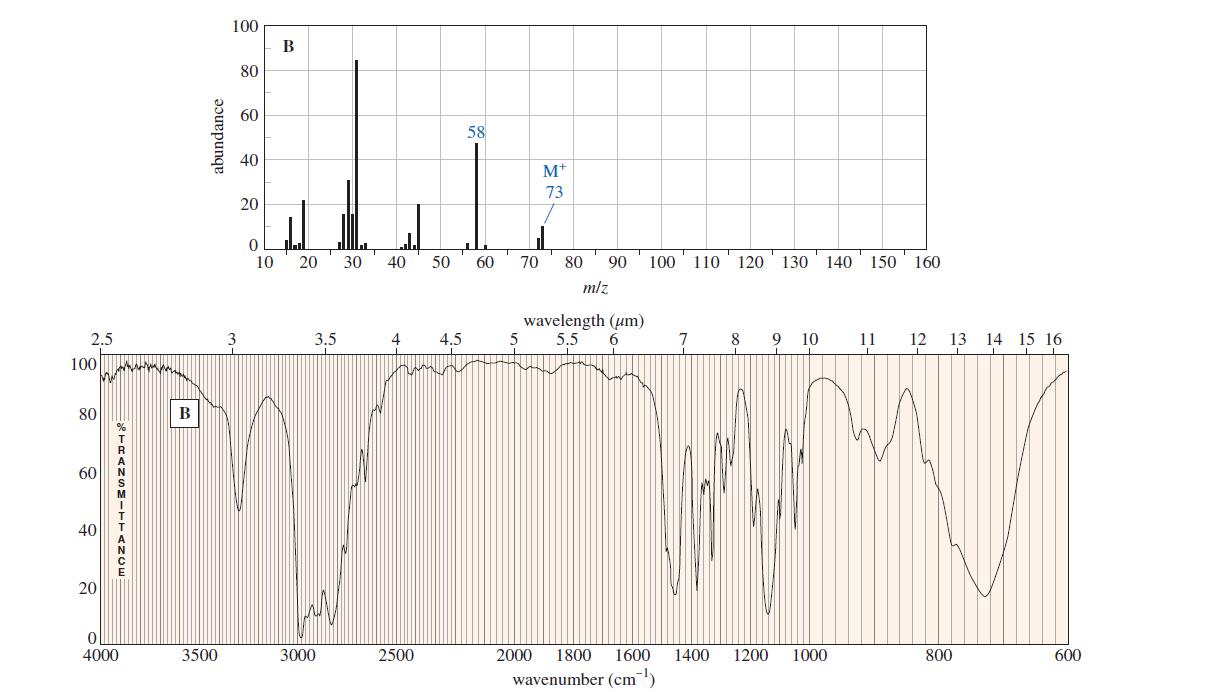
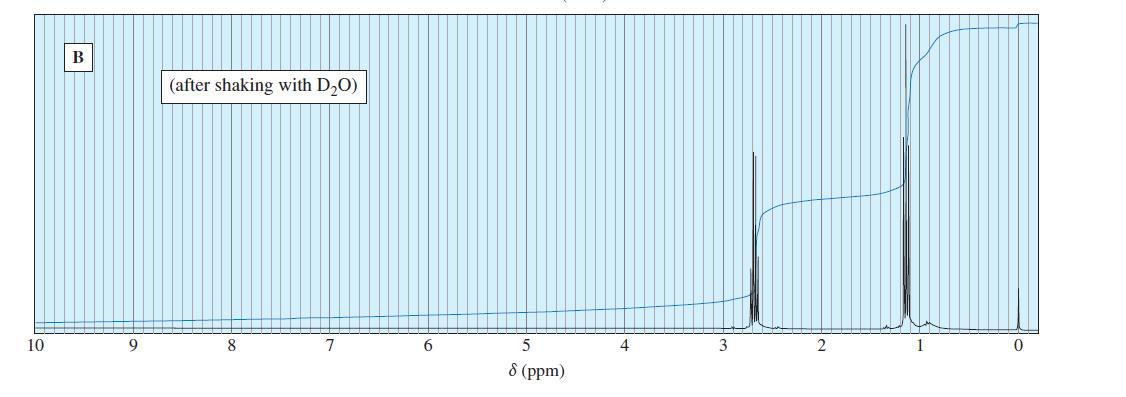
Question. The following spectra for A and B correspond to two structural isomers. The NMR singlet at1.16in spectrum A disappears when the sample is shaken with
. The singlet at
0.6 ppm in the spectrum B disappears on shaking with
. Propose structures for these isomers, and show how your structures correspond to the spectra. Show what cleavage is responsible for the base peak at
44 in the mass spectrum of A and the prominent peak at
58 in the mass spectrum of B.
Question: Several additional amine synthesies are effectively limited to making primry amines. The reduction of azides and nitro compounds and the Gabriel synthesis leave the carbon chain unchanged. Formation and reduction of a nitrile adds one carbon atom. Show how these amines syntheses can be used for the following conversions.
(a) allyI bromide → allylamine
(b) ethylbenzene → p-ethylaniline
(c) 1-bromo- 3-phenylheptane → 3-phenylheptan- 1-amine
(d) 1 -bromo- 3-phenylheptane → 4-phenyloctan- 1-anime
What do you think about this solution?
We value your feedback to improve our textbook solutions.