Chapter 21: 4P (page 817)
Give the IUPAC name for each aldehyde.
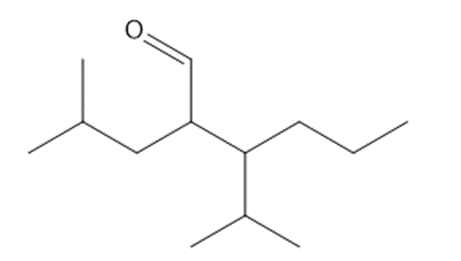
a.2-isobutyl-3-isopropylhexanal
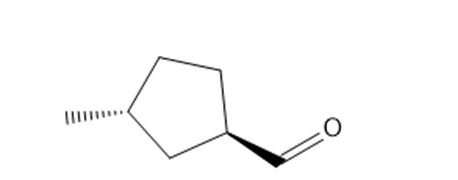
b. trans-3-methylcyclopentanecarbaldehyde
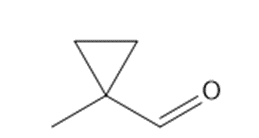
c. 1-methylcyclopropanecarbaldehyde
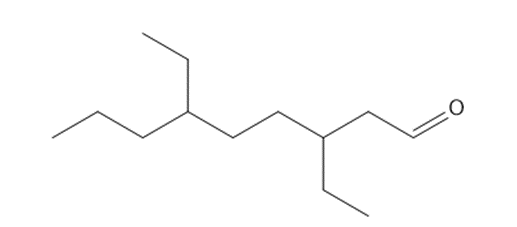
d. 3,6-diethylnonanal
Short Answer
a.
b
c
d
Learning Materials
Features
Discover
Chapter 21: 4P (page 817)
Give the IUPAC name for each aldehyde.
a.2-isobutyl-3-isopropylhexanal
b. trans-3-methylcyclopentanecarbaldehyde
c. 1-methylcyclopropanecarbaldehyde
d. 3,6-diethylnonanal
a.

b

c

d

All the tools & learning materials you need for study success - in one app.
Get started for free
Draw a stepwise mechanism for the following reaction, a key step in the synthesis ofticlopidine, a drug that inhibits platelet aggregation. Ticlopidine has been used to reduce therisk of stroke in patients who cannot tolerate aspirin.
Maltose is a carbohydrate present in malt, the liquid obtained from barley and other grains. Although maltose has numerous functional groups, its reactions are explained by the same principles we have already encountered.
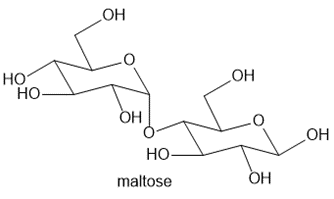
a. Label the acetal and hemiacetal carbons.b. What products are formed when maltose is treated with each of the following reagents: [1] ; [2] and HCl; [3] excess NaH, then excess ?c. Draw the products formed when the compound formed in Reaction [3] of part (b) is treated with aqueous acid.The reactions in parts (b) and (c) are used to determine structural features of carbohydrates like maltose. We will learn much more about maltose and similar carbohydrates in Chapter 28.
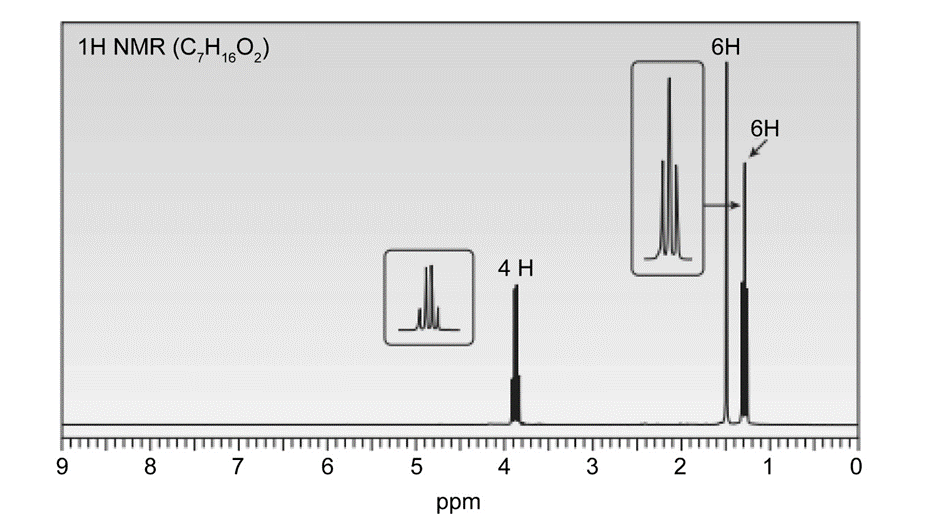
A solution of acetone (CH3)2C=O in ethanol CH3CH2OH in the presence of a trace of acid was allowed to stand for several days, and a new compound of molecular formula C7H16O2 was formed. The IR spectrum showed only one major peak in the functional group region around 3000 cm-1 , and the 1H-NMR spectrum is given here. What is the structure of the product?
Give the structure corresponding to each name:
(a) sec-butyl ethyl ketone
(b) methyl vinyl ketone
(c) p-ethylacetophenone
(d) 3-benzoyl-2-benzylcyclopentanone
(e) 6,6-dimethylcyclohex-2-enone
(f) 3-ethylhex-5-enal
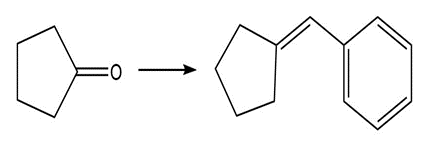
Show two methods to synthesize each alkene: a one-step method using a Wittig reagent, and a two-step method that forms a carbon–carbon bond with an organometallic reagent in one of the steps.
a.
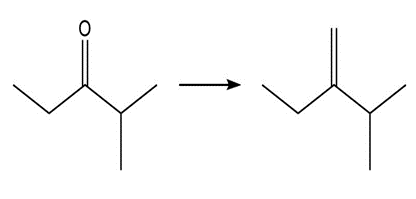
b.
What do you think about this solution?
We value your feedback to improve our textbook solutions.