Chapter 21: 30P (page 817)
Label each compound as an acetal, a hemiacetal, or an ether.
a.
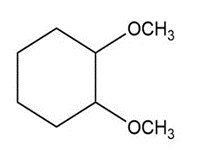
b.
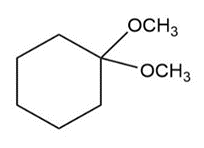
c.
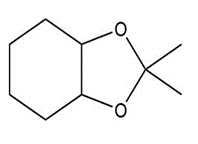
d.
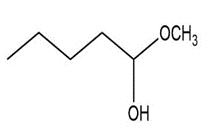
Short Answer
a. Ether
b. Open acetal
c. Cyclic acetal
d. Hemiacetal
Learning Materials
Features
Discover
Chapter 21: 30P (page 817)
Label each compound as an acetal, a hemiacetal, or an ether.
a.

b.

c.

d.

a. Ether
b. Open acetal
c. Cyclic acetal
d. Hemiacetal
All the tools & learning materials you need for study success - in one app.
Get started for free
Show two methods to synthesize each alkene: a one-step method using a Wittig reagent, and a two-step method that forms a carbon–carbon bond with an organometallic reagent in one of the steps.
a.
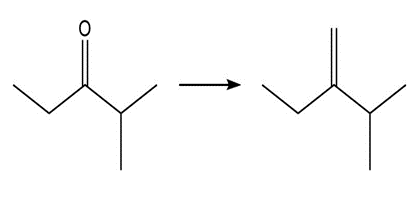
b.
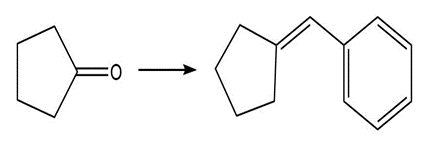
Which compound forms the higher concentration of hydrate at equilibrium, PhCOCHO or PhCH2CHO ? Explain your reasoning.
Compounds A and B have molecular formula C9H10O . Identify their structures from the 1HNMR and IR spectra given.
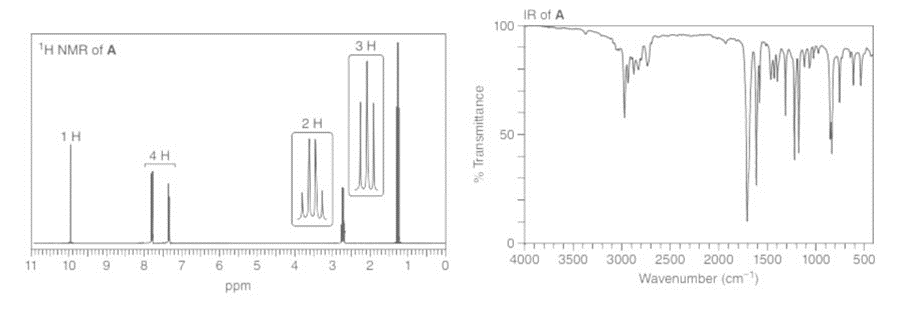
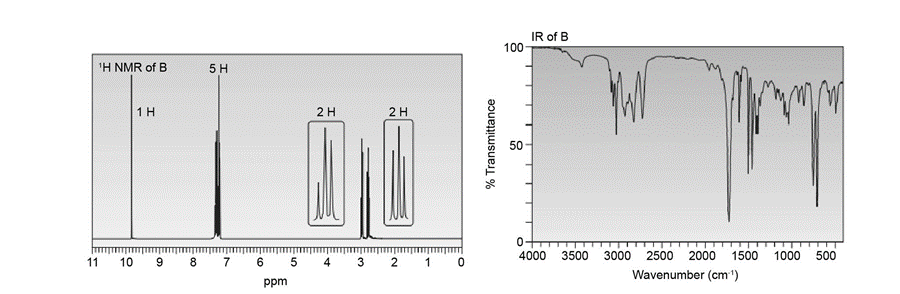
Draw a stepwise mechanism for the following reaction, a key step in the synthesis ofticlopidine, a drug that inhibits platelet aggregation. Ticlopidine has been used to reduce therisk of stroke in patients who cannot tolerate aspirin.
Draw the products of each reaction.
a.

b.
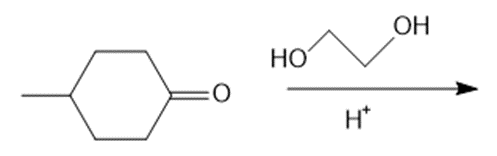
c.
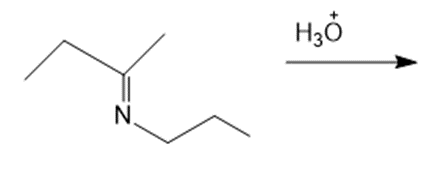
d.
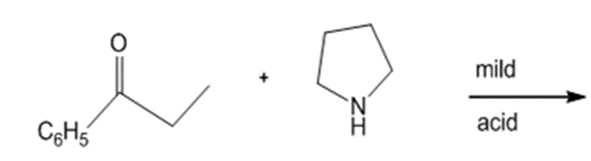
e.
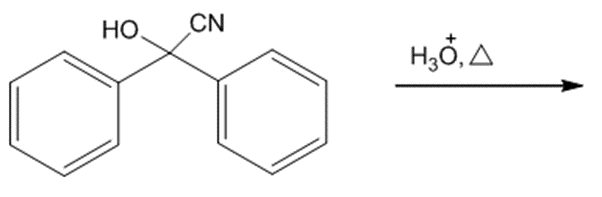
f.
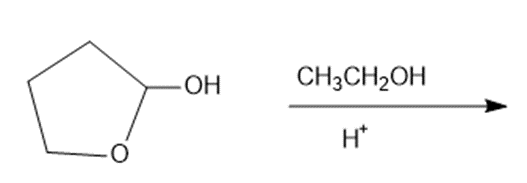
g.
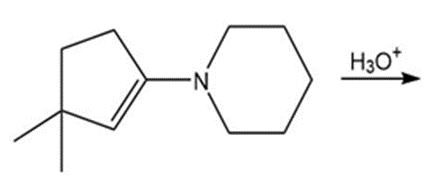
h.
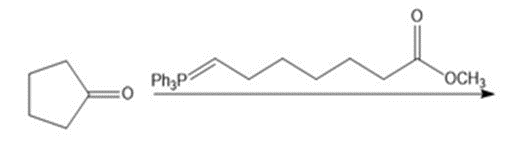
What do you think about this solution?
We value your feedback to improve our textbook solutions.