Chapter 4: Q8E (page 215)
Fill in the blank with a single chemical formula for a covalent compound that will balance the equation:
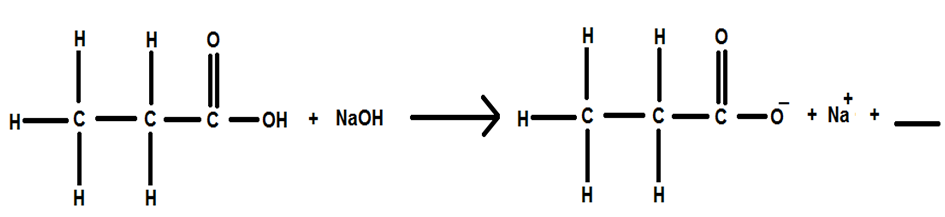
Short Answer
The complete balanced equation is represented as below:
Learning Materials
Features
Discover
Chapter 4: Q8E (page 215)
Fill in the blank with a single chemical formula for a covalent compound that will balance the equation:

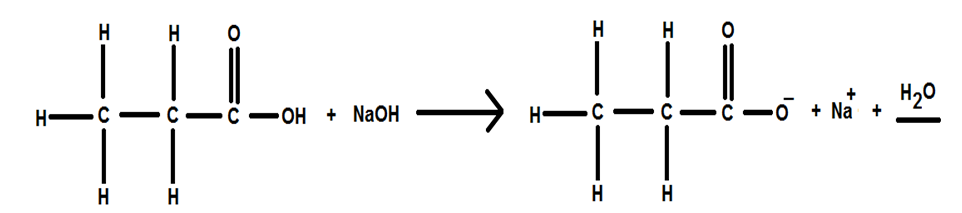
The complete balanced equation is represented as below:

All the tools & learning materials you need for study success - in one app.
Get started for free
Write the net ionic equation representing the neutralization of any strong acid with an ionic hydroxide.
(Hint: Consider the ions produced when a strong acid is dissolved in water)
Aqueous hydrogen fluoride (hydrofluoric acid) is used to etch glass and to analyze minerals for their silicon content. Hydrogen fluoride will also react with sand (silicon dioxide).
(a) Write an equation of the reaction of solid silicon dioxide with hydrofluoric acid to yield gaseous silicon tetrafluoride and liquid water.
(b)The mineral fluorite (calcium fluoride) occurs extensively in Illinois. Solid calcium fluoride can also be prepared by the reaction of aqueous solution of calcium chloride and sodium fluoride, yielding aqueous sodium chloride as the other product. Write complete and net ionic equations for this reaction.
What is the percent yield of a reaction that produces 12.5g of the gas freon CF2Cl2 from 32.9 g of CCl4 and excess HF.
\(CC{l_4} + 2HF \to C{F_2}\)
Complete and balance each of the following oxidation-reduction reactions,such that it results in the highest possible oxidation state for the oxidized atoms.
(a) \(Al\left( s \right) + {F_2}\left( g \right) \to \)
(b) \(Al\left( s \right) + CuB{r_2}\left( {aq} \right) \to \) (single displacement)
(c) \({P_4}\left( s \right) + {O_2}\left( g \right) \to \)
(d) \(Ca\left( s \right) + {H_2}O\left( l \right) \to \) (products are a strong base and a diatomic gas)
What do you think about this solution?
We value your feedback to improve our textbook solutions.