Chapter 4: Q13E (page 216)
Indicate what type or types of reaction each of the following represents:
(a) \(\)\(Ca\left( s \right) + B{r_2}\left( l \right) \to CaB{r_2}\left( s \right)\)
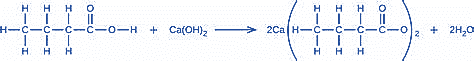
(b) \(Ca{\left( {OH} \right)_2} + 2HBr\left( {aq} \right) \to CaB{r_2}\left( {aq} \right) + 2{H_2}O\left( l \right)\)
(c) \({C_6}{H_{12}}\left( l \right) + 9{O_2}\left( g \right) \to 6C{O_2}\left( g \right) + 6{H_2}O\left( l \right)\)\(\)
Short Answer
(a) Oxidation-reduction reaction
(b) Acid-base reaction
(c) Oxidation-reduction reaction (combustion)