Chapter 4: 4.5 CYL (page 191)
Assign oxidation state to elements whose atoms are underlined in each of the following compounds or ions:
- KNO3
- AlH3
- NH4+
- H2PO4-
Short Answer
The respective oxidation states are:
- N = +5
- Al = +3
- N = -3
- P = +5
Learning Materials
Features
Discover
Chapter 4: 4.5 CYL (page 191)
Assign oxidation state to elements whose atoms are underlined in each of the following compounds or ions:
The respective oxidation states are:
All the tools & learning materials you need for study success - in one app.
Get started for free
Silver can be separated from gold because silver dissolves in nitric acid while gold does not. Is the dissolution of silver in nitric acid an acid-base reaction or an oxidation-reduction reaction? Explain your answer.
In a laboratory experiment, the reaction of 3.0 mol of\({H_2}\)with 2.0 mol of\({I_2}\)produced 1.0 mol of HI. Determine the theoretical yield in grams and the percent yield for this reaction.
What mass of gallium oxide Ga2O3 can be prepared from 29 g of gallium metal? The equation for the reaction is \(4Ga + 3{O_2} \to 2G{a_2}{O_3}\)
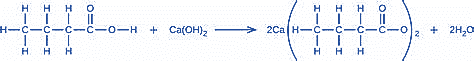
What mass of Ca(OH)2 will react with 25.0 g of propionic acid to form the preservative calcium propionate according to the equation?
What is the limiting reactant when 1.50 g of lithium and 1.50 g of nitrogen combine to form lithium nitride, a component of advanced batteries, according to the following unbalanced equation?\(Li + {N_2} \to L{i_3}N\).
What do you think about this solution?
We value your feedback to improve our textbook solutions.