Chapter 11: Q35 E (page 648)
What is the difference between a 1 M solution and a 1 m solution?
Short Answer
1M solution contains 1mole of solute for every 1L solution whereas 1m solution contains 1mole of solute for every kg of solvent.
Learning Materials
Features
Discover
Chapter 11: Q35 E (page 648)
What is the difference between a 1 M solution and a 1 m solution?
1M solution contains 1mole of solute for every 1L solution whereas 1m solution contains 1mole of solute for every kg of solvent.
All the tools & learning materials you need for study success - in one app.
Get started for free
Indicate the most important type of intermolecular attraction responsible for solvation in each of the following solutions:
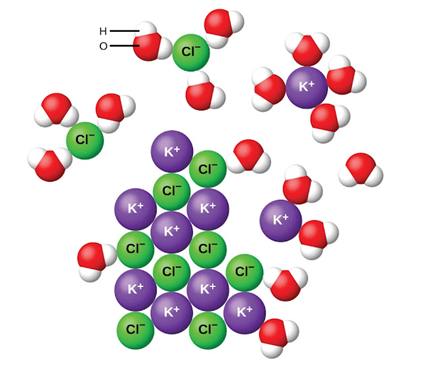
2. Methanol,\({\bf{CH}}3{\bf{OH}}\), dissolved in ethanol, \({\bf{C}}2{\bf{H}}5{\bf{OH}}\)
3. Methane,\({\bf{C}}{{\bf{H}}_{\bf{4}}}\), dissolved in benzene, \({{\bf{C}}_{\bf{6}}}{{\bf{H}}_{\bf{6}}}\)
4. The polar halocarbon \({\bf{C}}{{\bf{F}}_{\bf{2}}}{\bf{C}}{{\bf{l}}_{\bf{2}}}\)dissolved in the polar halocarbon \({\bf{CF}}2{\bf{ClCFCl2}}\)
5. \({{\bf{O}}_{\bf{2}}}\left( {\bf{l}} \right)\)in \({{\bf{N}}_{\bf{2}}}\left( {\bf{l}} \right){\bf{.}}\) \(\)
What is the freezing point of a solution of 9.04 g of\({\bf{I_2}}\)in 75.5 g of benzene?
Compare the processes that occur when methanol (\({\bf{CH}}_3{\bf{OH}}\)), hydrogen chloride (\({\bf{HCl}}\)), and sodium hydroxide (\({\bf{NaOH}}\)) dissolve in water. Write equations and prepare sketches showing the form in which each of these compounds is present in its respective solution.
Which is/are part of the macroscopic domain of solutions and which is/are part of the microscopic domain: boiling point elevation, Henry’s law, hydrogen bond, ion-dipole attraction, molarity, nonelectrolyte, non-stoichiometric compound, osmosis, solvated ion?
Lysozyme is an enzyme that cleaves cell walls. A 0.100 L sample of a solution of lysozyme that contains 0.0750 g of the enzyme exhibits an osmotic pressure of \({\bf{1}}{\bf{.32 \times 1}}{{\bf{0}}^{{\bf{ - 3}}}}\) atm at 25 °C. What is the molar mass of lysozyme?
What do you think about this solution?
We value your feedback to improve our textbook solutions.