Chapter 20: Q52E (page 1145)
Yields in organic reactions are sometimes low. What is the percent yield of a process that produces 13.0 g of ethyl acetate from \(10.0\;g\) of\(C{H_3}C{O_2}H\) ?
Short Answer
The yield is 93 %.
Learning Materials
Features
Discover
Chapter 20: Q52E (page 1145)
Yields in organic reactions are sometimes low. What is the percent yield of a process that produces 13.0 g of ethyl acetate from \(10.0\;g\) of\(C{H_3}C{O_2}H\) ?
The yield is 93 %.
All the tools & learning materials you need for study success - in one app.
Get started for free
Give the complete IUPAC name and the common name for each of the following compounds:
a)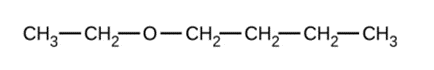
b)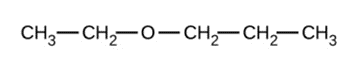
c)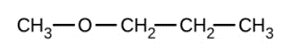
What is the molecular structure about the nitrogen atom in trimethyl amine and in the trimethyl ammonium ion, \({\left( {C{H_3}} \right)_3}N{H^ + }\)? What is the hybridization of the nitrogen atom in trimethyl amine and in the trimethyl ammonium ion?
Write Lewis structures and describe the molecular geometry of each carbon atom in the following compounds:
(a) cis-3-hexene
(b) cis-1-chloro-2-bromoethene
(c) 2-pentyne
(d)trans-6-ethyl-7-methyl-2-octene
Write two complete balanced equations for each of the following reactions, one using condensed formulas and one using Lewis structures.
(a) propanol is converted to dipropyl ether
(b) propene is treated with water in dilute acid.
Predict the products of oxidizing the molecules shown in this problem. In each case, identify the product that will result from the minimal increase in oxidation state for the highlighted carbon atom:
a)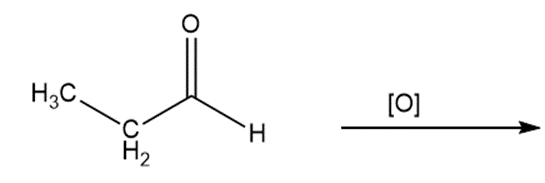
b)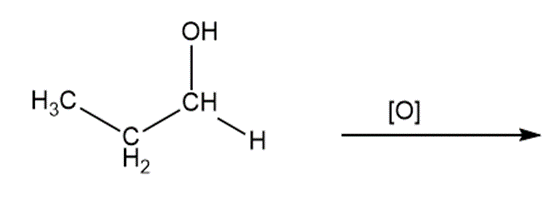
c)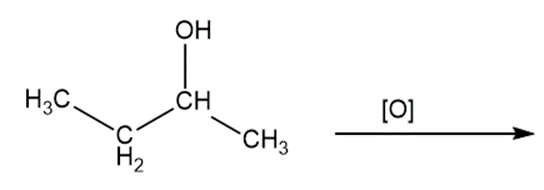
What do you think about this solution?
We value your feedback to improve our textbook solutions.