Chapter 10: Q10.10CYL (page 547)
What is the total amount of heat released when 94.0 g water at 80.0 °C cools to form ice at −30.0 °C?
Short Answer
Total amount of heat released when water cools to form ice is found \(68.7{\rm{KJ}}\)
Learning Materials
Features
Discover
Chapter 10: Q10.10CYL (page 547)
What is the total amount of heat released when 94.0 g water at 80.0 °C cools to form ice at −30.0 °C?
Total amount of heat released when water cools to form ice is found \(68.7{\rm{KJ}}\)
All the tools & learning materials you need for study success - in one app.
Get started for free
From the phase diagram for carbon dioxide determine the state of CO2 at:
\(\begin{aligned}{\left( {\rm{a}} \right){\rm{ 20 ^\circ C and 1000 kPa}}}\\{\left( {\rm{b}} \right){\rm{ 10 ^\circ C and 2000 kPa}}}\\{\left( {\rm{c}} \right){\rm{ 10 ^\circ C and 100 kPa}}}\\{\left( {\rm{d}} \right){\rm{ - 40 ^\circ C and 500 kPa}}}\\{\left( {\rm{e}} \right){\rm{ - 80 ^\circ C and 1500 kPa}}}\\{\left( {\rm{f}} \right){\rm{ - 80 ^\circ C and 10 kPa}}}\end{aligned}\)
Elemental carbon has one gas phase, one liquid phase, and two different solid phases, as shown in the phase diagram
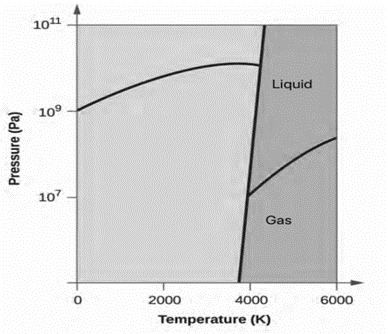
The types of intermolecular forces in a substance are identical whether it is a solid, a liquid, or a gas. Why then does a substance change phase from a gas to a liquid or to a solid?
Barium crystallizes in a body-centered cubic unit cell with an edge length of \(5.025{A^o}\)
(a) What is the atomic radius of barium in this structure?
(b) Calculate the density of barium
The density of aluminium is \(2.7\;{\rm{g}}/{\rm{c}}{{\rm{m}}^3}\); that of silicon is \(2.3\;{\rm{g}}/{\rm{c}}{{\rm{m}}^3}\). Explain why Si has the lower density even though it has heavier atoms.
What do you think about this solution?
We value your feedback to improve our textbook solutions.