Chapter 12: Q77E. (page 712)
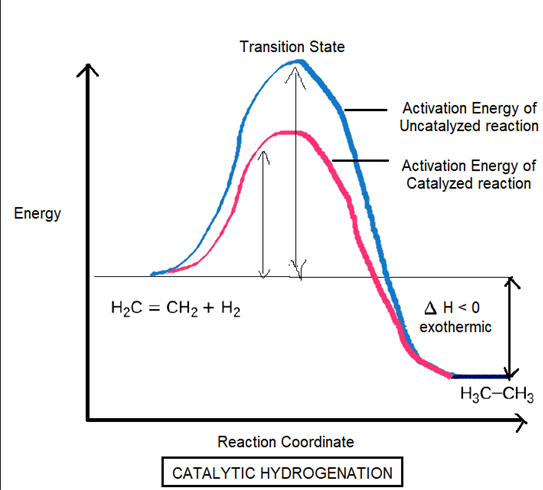
Account for the increase in reaction rate brought about by a catalyst.
Short Answer
Catalysts increases the rate of the reaction by decreasing the activation energy, by involving another reaction mechanism by allowing the reaction to attain equilibrium faster.