Chapter 13: Q82E (page 760)
Question: Calculate the equilibrium concentrations that result when 0.25 M O2 and 1.0 M HCl react and come to equilibrium.
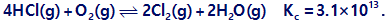
Short Answer
The equilibrium concentration is

Learning Materials
Features
Discover
Chapter 13: Q82E (page 760)
Question: Calculate the equilibrium concentrations that result when 0.25 M O2 and 1.0 M HCl react and come to equilibrium.
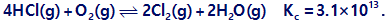
The equilibrium concentration is

All the tools & learning materials you need for study success - in one app.
Get started for free
Assume that the change in pressure of \({H_2}S\) is small enough to be neglected in the following problem.(a) Calculate the equilibrium pressures of all species in an equilibrium mixture that results from the decomposition of H2S with an initial pressure of 0.824 atm.
\(2{H_2}S(g) \rightleftharpoons 2{H_2}(g) + {S_2}(g)\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,{K_p} = 2.2 \times {10^{( - 6)}}\)
(b) Show that the change is small enough to be neglected.
Question: A 1.00-L vessel at 400 °C contains the following equilibrium concentrations: N2, 1.00M; H2, 0.50M; and NH3, 0.25M. How many moles of hydrogen must be removed from the vessel to increase the concentration of nitrogen to 1.1M?
Question: Butane exists as two isomers, n−butane and isobutane.
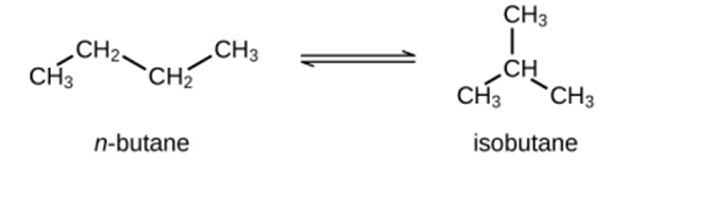
\({K_P} = 2.5\;at\;2{5^o}C\)
What is the pressure of isobutane in a container of the two isomers at equilibrium with a total pressure of 1.22 atm?
The following reaction has \({K_P} = 4.50 \times {10^{ - 5}}\) at \(720\;{\rm{K}}\).
\({{\rm{N}}_2}(g) + 3{{\rm{H}}_2}(g) \rightleftharpoons 2{\rm{N}}{{\rm{H}}_3}(g)\)
If a reaction vessel is filled with each gas to the partial pressures listed, in which direction will it shift to reach equilibrium?
\(P\left( {{\rm{N}}{{\rm{H}}_3}} \right) = 93\;{\rm{atm}},\;P\left( {\;{{\rm{N}}_2}} \right) = 48\;{\rm{atm}},\;{\rm{and}}\;P\left( {{{\rm{H}}_2}} \right) = 52\)
A sample of ammonium chloride was heated in a closed container. NH4 Cl (s)⇌ NH3 (g) + HCl(g)at equilibrium, the pressure of NH3 (g)was found to be 1.75 atm. What is the value of the equilibrium constant, Kp, for the decomposition at this temperature?
What do you think about this solution?
We value your feedback to improve our textbook solutions.