Chapter 13: Q33 E (page 752)
What property of a reaction can we use to predict the effect of a change in temperature on the value of an equilibrium constant?
Short Answer
Changes in enthalpy may be used.
Learning Materials
Features
Discover
Chapter 13: Q33 E (page 752)
What property of a reaction can we use to predict the effect of a change in temperature on the value of an equilibrium constant?
Changes in enthalpy may be used.
All the tools & learning materials you need for study success - in one app.
Get started for free
Question: In a 3.0-L vessel, the following equilibrium partial pressures are measured: \({{\rm{N}}_2}\),190 torr;\({{\rm{H}}_2}\), 317 torr;\({\rm{N}}{{\rm{H}}_3}\)\(1.00 \times {10^3}\)torr.
Question: Butane exists as two isomers, n−butane and isobutane.
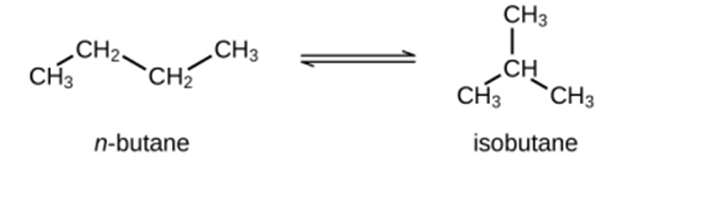
\({K_P} = 2.5\;at\;2{5^o}C\)
What is the pressure of isobutane in a container of the two isomers at equilibrium with a total pressure of 1.22 atm?
Question: The hydrolysis of the sugar sucrose to the sugars glucose and fructose follows a first-order rate equation for the disappearance of sucrose.
C12 H22 O11(aq) + H2��(��)⟶C6 H12 O6 (aq) + C6 H12 O6 (aq)
Rate = k[C12H22O11]
In neutral solution, k = 2.1 × 10−11/s at 27 °C. (As indicated by the rate constant, this is a very slow reaction. In the human body, the rate of this reaction is sped up by a type of catalyst called an enzyme.) (Note: That is not a mistake in the equation—the products of the reaction, glucose and fructose, have the same molecular formulas, C6H12O6, but differ in the arrangement of the atoms in their molecules). The equilibrium constant for the reaction is 1.36 × 105 at 27 °C. What are the concentrations of glucose, fructose, and sucrose after a 0.150 M aqueous solution of sucrose has reached equilibrium? Remember that the activity of a solvent (the effective concentration) is 1.
Analysis of the gases in a sealed reaction vessel containing \(N{H_3}\), \({N_2}\), and \({H_2}\) at equilibrium at \(40{0^0}C\) established the concentration of \({N_2}\) to be \(1.2M\) and the concentration of \({H_2}\) to be \(0.24M\).
\({N_2}(g) + 3{H_2}(g) \rightleftharpoons 2N{H_3}(g)\)
\({K_c} = 0.50\,at\,40{0^o}C\)
Calculate the equilibrium molar concentration of \(N{H_3}\).
Round the following to the indicated number of significant figures:
(a) 0.424 (to two significant figures)
(b) 0.0038661 (to three significant figures)
(c) 421.25 (to four significant figures)
(d) 28,683.5 (to five significant figures)
What do you think about this solution?
We value your feedback to improve our textbook solutions.