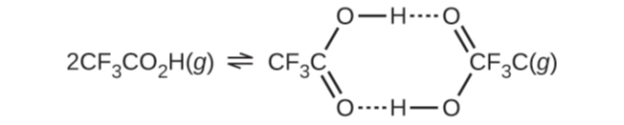
Chapter 13: Q22E (page 752)
Which of the systems described in Exercise 13.16 give homogeneous equilibria? Which give heterogeneous equilibria?
(a) \({N_2}(g) + 3{H_2}(g)\rightleftharpoons 2N{H_3}(g)\)
(b) \(4N{H_3}(g) + 5{O_2}(g)\rightleftharpoons 4NO(g) + 6{H_2}O(g)\)
(c) \({N_2}{O_4}(g)\rightleftharpoons 2N{O_2}(g)\)
(d) \(C{O_2}(g) + {H_2}(g)\rightleftharpoons CO(g) + {H_2}O(g)\)
(e) \(N{H_4}Cl(s)\rightleftharpoons N{H_3}(g) + HCl(g)\)
(f) \(2\;Pb{\left( {N{O_3}} \right)_2}(s)\rightleftharpoons 2PbO(s) + 4N{O_2}(g) + {O_2}(g)\)
(g) \(2{H_2}(g) + {O_2}(g)\rightleftharpoons 2{H_2}O(l)\)
(h) \({S_8}(g)\rightleftharpoons 8\;S(g)\)
Short Answer
- The system is Homogenous.
- The system is Homogenous.
- The system is Homogenous.
- The system is homogenous.
- The system is Heterogenous.
- The system is heterogeneous.
- The system is Heterogenous.
- The system is homogenous.