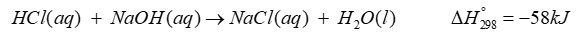Chapter 1: Q5.3-50E (page 9)
How much heat is produced when 100 mL of 0.250 M HCl (density, 1.00 g/mL) and 200 mL of 0.150 M NaOH (density, 1.00 g/mL) are mixed?
\({\bf{HCl(aq) + NaOH(aq)}} \to {\bf{NaCl(aq) + }}{{\bf{H}}_{\bf{2}}}{\bf{O(l) \Delta H}}_{{\bf{298}}}^{\bf{^\circ }}{\bf{ = - 58kJ}}\)
If both solutions are at the same temperature and the heat capacity of the products is 4.19 J/g °C, how much will the temperature increase? What assumption did you make in your calculation?
Short Answer
The amount of energy released in the solution will be 1.45 kJ and the change in temperature in the later part of the problem will be \(1.15^\circ C\).