Chapter 15: Q40 E (page 873)
A volume of 0.800 L of a 2×10-4-MBa(NO3)2 solution is added to 5×10-4 MLi2 SO4. Does BaSO4 precipitate? Explain your answer.
Short Answer
Ki is smaller than Ksp for BaSO4, no precipitation of BaSO4 will occur
Learning Materials
Features
Discover
Chapter 15: Q40 E (page 873)
A volume of 0.800 L of a 2×10-4-MBa(NO3)2 solution is added to 5×10-4 MLi2 SO4. Does BaSO4 precipitate? Explain your answer.
Ki is smaller than Ksp for BaSO4, no precipitation of BaSO4 will occur
All the tools & learning materials you need for study success - in one app.
Get started for free
Question: How many grams of \(Pb{(OH)_2}\)will dissolve in 500 mL of a \(0.050 - MPbC{l_2}\;solution\;\left( {{K_{sp}} = 1.2 \times 1{0^{ - 15}}} \right)?\)
What mass of NaCN must be added to 1L of 0.010MMg (NO3)2in order to produce the first trace of Mg(OH)2?
What is the molar solubility of \({\bf{Tl}}{\left( {{\bf{OH}}} \right)_{\bf{3}}}\) in a \({\bf{0}}.{\bf{10}}{\rm{ }}{\bf{M}}\) solution of \({\bf{N}}{{\bf{H}}_{\bf{3}}}\) ?
What is the molar solubility of CaF2 in a 0.100 M solution of HF? Kafor HF = 7.2 × 10–4.
Question: Hydrogen sulfide is bubbled into a solution that is 0.10 M in both \(P{b^{2 + }}\)and \(F{e^{2 + }}\)and 0.30 M in HCl. After the solution has come to equilibrium it is saturated with \({H_2}S\) ((\({H_2}S\)) = 0.10 M). What concentrations of \(P{b^{2 + }}\)and \(F{e^{2 + }}\)remain in the solution? For a saturated solution of \({H_2}S\)we can use the equilibrium:
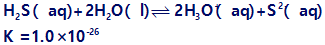
What do you think about this solution?
We value your feedback to improve our textbook solutions.