Chapter 7: Question 44 E (page 397)
Write resonance forms describing the distribution of electrons in each molecule or ion.
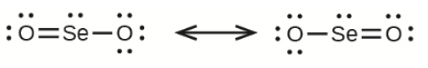
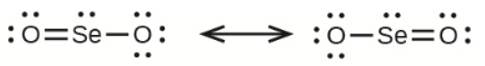
a) selenium dioxide, \({\rm{OSeO}}\)
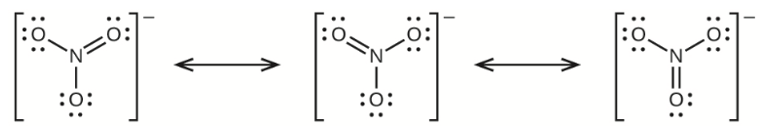
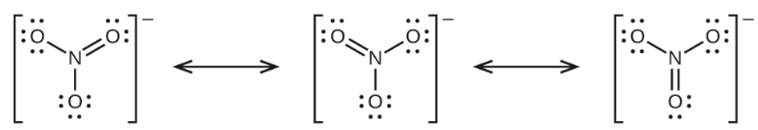
(b) nitrate ion, \({\rm{NO}}_{\rm{3}}^{\rm{ - }}\)
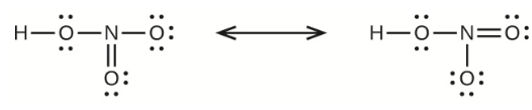
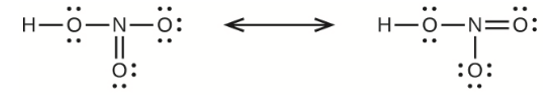
(c) nitric acid, \({\rm{HN}}{{\rm{O}}_{\rm{3}}}\) (\({\rm{N}}\) is bonded to an \({\rm{OH}}\) group and two \({\rm{O}}\) atoms)
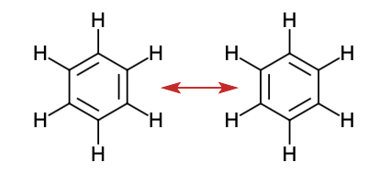
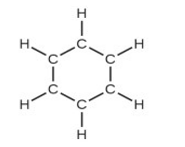
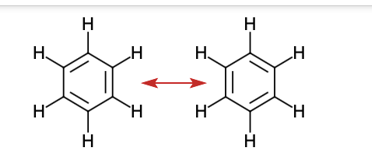
(d) benzene, \({{\rm{C}}_{\rm{6}}}{{\rm{H}}_{\rm{6}}}\):
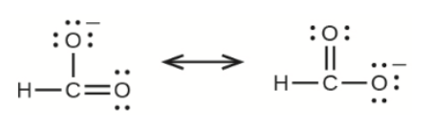
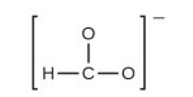
(e) the formate ion:
Short Answer
The resonance forms for the following compounds are as follows –
(a) Selenium dioxide -
(b) Nitrate Ion –
(c) Nitric Acid –
(d) Benzene –
(e) Formate Ion –