In a\({\rm{100}}{\rm{.0 g}}\)sample, there are\({\rm{85}}{\rm{.7 g C}}\) and\({\rm{14}}{\rm{.3 g H}}\).
Now we must compute the mole of\({\rm{C}}\) and\({\rm{H}}\), which we shall accomplish by dividing their mass by their molecular weight, as follows:-
The moles of carbon is:
\(\dfrac{{{\rm{85}}{\rm{.7 g}}}}{{{\rm{12}}{\rm{.001 g mo}}{{\rm{l}}^{{\rm{ - 1}}}}}}{\rm{ = 7}}{\rm{.14 mol C}}\)
The moles of hydrogen is:
\(\dfrac{{{\rm{14}}{\rm{.3 g}}}}{{{\rm{1}}{\rm{.0079 g mo}}{{\rm{l}}^{{\rm{ - 1}}}}}}{\rm{ = 14}}{\rm{.19 mol H}}\)
To compute the formula, divide the mole by the smallest mole:-
\(\dfrac{{{\rm{7}}{\rm{.14 mol}}}}{{{\rm{7}}{\rm{.14 mol}}}}{\rm{ = 1 C}}\)
\(\dfrac{{{\rm{14}}{\rm{.19 mol}}}}{{{\rm{7}}{\rm{.14 mol}}}}{\rm{ = 2 H}}\)
The formula is\({\rm{C}}{{\rm{H}}_{\rm{2}}}\)having the molecular mass as\({\rm{14}}\).
But, here, the molecular mass of the compound is\({\rm{42}}\).
So, the correct formula will be\({\rm{3 \times C}}{{\rm{H}}_{\rm{2}}}\) or \({{\rm{C}}_{\rm{3}}}{{\rm{H}}_{\rm{6}}}\) .
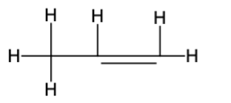
The Lewis structure is as follows:-
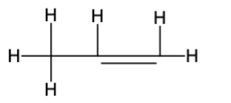
Therefore, the formula is\({{\rm{C}}_{\rm{3}}}{{\rm{H}}_{\rm{6}}}\) and the Lewis structure is: