Chapter 7: Q73E (page 400)
Question: Complete the following Lewis structure by adding bonds (not atoms), and then indicate the longest bond:
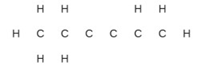
Short Answer
The Lewis Structure with longest \({\rm{C - C}}\) bond is as follows –
Learning Materials
Features
Discover
Chapter 7: Q73E (page 400)
Question: Complete the following Lewis structure by adding bonds (not atoms), and then indicate the longest bond:
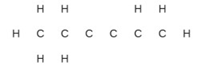
The Lewis Structure with longest \({\rm{C - C}}\) bond is as follows –

All the tools & learning materials you need for study success - in one app.
Get started for free
Which compound in each of the following pairs has the larger lattice energy? Note: \({\rm{M}}{{\rm{g}}^{{\rm{2 + }}}}\) and \({\rm{L}}{{\rm{i}}^{\rm{ + }}}\) have similar radii; \({{\rm{O}}^{{\rm{2 - }}}}\) and \({{\rm{F}}^{\rm{ - }}}\) have similar radii. Explain your choices.
(a) \({\rm{MgO}}\) or \({\rm{MgSe}}\)
(b) \({\rm{LiF}}\) or \({\rm{MgO}}\)
(c) \({\rm{L}}{{\rm{i}}_{\rm{2}}}{\rm{O}}\) or \({\rm{LiCl}}\)
(d) \({\rm{L}}{{\rm{i}}_{\rm{2}}}{\rm{Se}}\) or \({\rm{MgO}}\)
Question: Calculate the formal charge of each element in the following compounds and ions:
(a) \({{\rm{F}}_{\rm{2}}}{\rm{CO}}\)
(b) \({\rm{N}}{{\rm{O}}^{\rm{ - }}}\)
(c) \({\rm{BF}}_{\rm{4}}^{\rm{ - }}\)
(d) \({\rm{SnCl}}_{\rm{3}}^{\rm{ - }}\)
(e) \({{\rm{H}}_{\rm{2}}}{\rm{CC}}{{\rm{H}}_{\rm{2}}}\)
(f) \({\rm{Cl}}{{\rm{F}}_{\rm{3}}}\)
(g) \({\rm{Se}}{{\rm{F}}_{\rm{6}}}\)
(h) \({\rm{PO}}_{\rm{4}}^{{\rm{3 - }}}\)
Which compound in each of the following pairs has the larger lattice energy? Note: \({\rm{B}}{{\rm{a}}^{{\rm{2 + }}}}\) and \({{\rm{K}}^{\rm{ + }}}\) have similar radii; \({{\rm{S}}^{{\rm{2 - }}}}\) and \({\rm{C}}{{\rm{l}}^{\rm{ - }}}\) have similar radii. Explain your choices.
(a) \({{\rm{K}}_{\rm{2}}}{\rm{O}}\) or \({\rm{N}}{{\rm{a}}_{\rm{2}}}{\rm{O}}\)
(b) \({{\rm{K}}_{\rm{2}}}{\rm{S}}\) or \({\rm{BaS}}\)
(c) \({\rm{KCl}}\) or \({\rm{BaS}}\)
(d) \({\rm{BaS}}\) or \({\rm{BaC}}{{\rm{l}}_{\rm{2}}}\)
Use the Molecule Shape simulator (http://openstaxcollege.org/l/16MolecShape) to explore real molecules. On the Real Molecules tab, select H2O. Switch between the “real” and “model” modes. Explain the difference observed.
Draw the Lewis structures and predict the shape of each compound or ion:
(a) \({\rm{C}}{{\rm{O}}_{\rm{2}}}\)
(b) \({\rm{NO}}_{\rm{2}}^{\rm{ - }}\)
(c) \({\rm{S}}{{\rm{O}}_{\rm{3}}}\)
(d) \({\rm{S}}{{\rm{O}}_{\rm{3}}}^{{\rm{2 - }}}\)
What do you think about this solution?
We value your feedback to improve our textbook solutions.