Chapter 14: Q14.3-39 E (page 829)
Which is the stronger base, \({\left( {C{H_3}} \right)_3}N\)or\({H_2}BO_3^ - \)?
Short Answer
\(\left( {C{H_3}} \right)N\) is the stronger base.
Learning Materials
Features
Discover
Chapter 14: Q14.3-39 E (page 829)
Which is the stronger base, \({\left( {C{H_3}} \right)_3}N\)or\({H_2}BO_3^ - \)?
\(\left( {C{H_3}} \right)N\) is the stronger base.
All the tools & learning materials you need for study success - in one app.
Get started for free
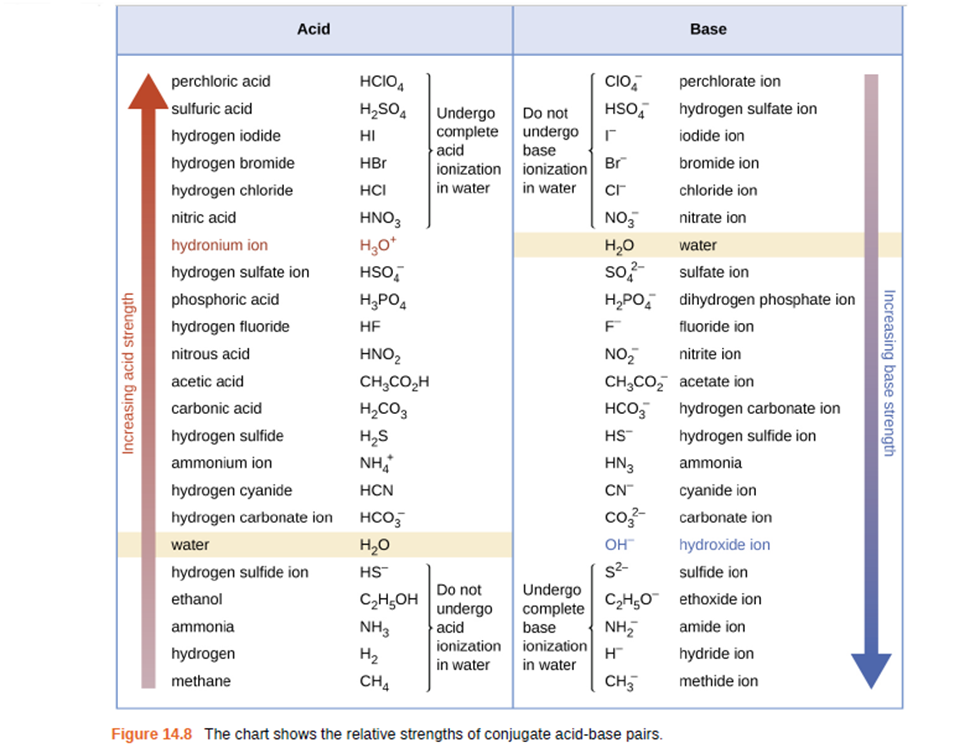
Using the \({K_a}\) value of \(1.4 \times 1{0^{ - 5}}\), place \(Al\left( {{H_2}O} \right)_6^{3 + }\) in the correct location in Figure 14.8
Question: Show by suitable net ionic equations that each of the following species can act as a Bronsted-Lowry base: (a) \({H_2}O\) (b) \(O{H^ - }\)(c) \(N{H_3}\)(d) \(C{N^ - }\)(e) \({S^{2 - }}\)(f) \({H_2}PO_4^ - \)
What are the\(pH\;and\;pOH\) of a solution of \(2.0\)\(M\)\(HCl\), which ionizes completely?
Why can we ignore the contribution of water to the concentrations of\({H_3}{O^ + }\)in the solutions of following acids:\(0.0092MHClO\), a weak acid\(0.0810MHCN\), a weak acid but not the contribution of water to the concentration of\(O{H^ - }\)?
What is the ionization constant at 25oC for the weak acid \(C{H_3}NH_3^ + \), the conjugate acid of the weak base \(C{H_3}N{H_2}\)\({K_b} = 4.4 \times 1{0^{ - 4}}\) ,.
What do you think about this solution?
We value your feedback to improve our textbook solutions.