Consider the figure,
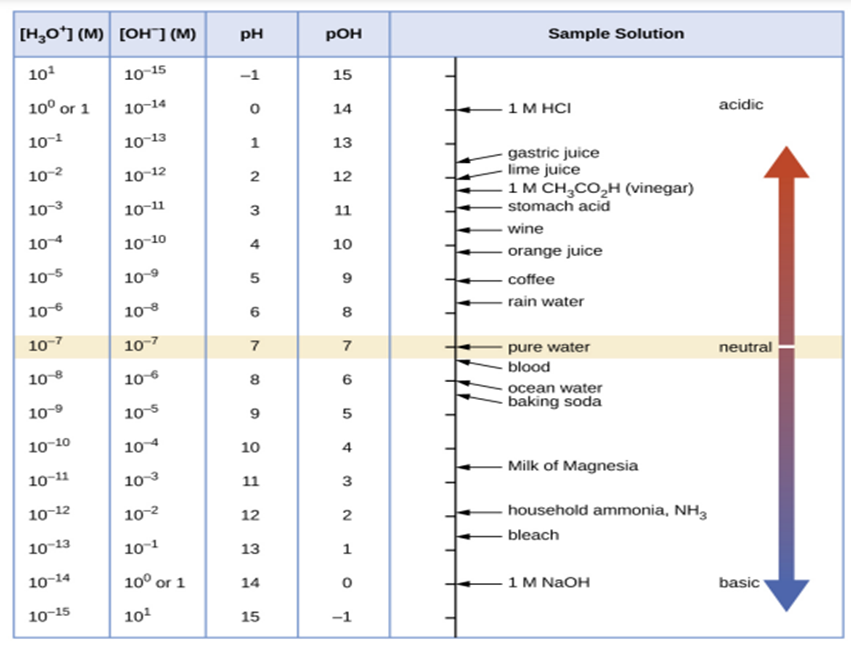
Hence, from the figure, \(pH\)of a wine is approximately\(3.5\).
Calculate the concentration of hydronium ions,
\(c\left( {{H_3}{O^ + }} \right) = {10^{ - pH}}mol{L^{ - 1}}\)
Substitute\(pH = 3.5\)
\(\begin{aligned}{c\left( {{H_3}{O^ + }} \right) = {{10}^{ - 3.5}}mol{L^{ - 1}}}\\{c\left( {{H_3}{O^ + }} \right) = 3.2 \cdot {{10}^{ - 4}}mol{L^{ - 1}}}\end{aligned}\)
Find the concentration of hydroxide ion using the equation,
\(c\left( {O{H^ - }} \right) = {10^{ - pOH}}mol{L^{ - 1}}\).
Calculate \(pOH\)using the equation,
\(pH + pOH = p{K_w}\)
Since,
\(p{K_w} = - \log {K_w}\)
Substitute, the ionization constant of water\(({K_w}) = 1.0 \times {10^{ - 14}}\)
\(\begin{aligned}{p{K_w} = - \log \left( {1.0 \cdot {{10}^{ - 14}}} \right)}\\{p{K_w} = 14}\end{aligned}\)
Thus, \(pOH\)of wine is,
\(\begin{aligned}{pOH = p{K_w} - pH}\\{pOH = 14 - 3.5}\\{pOH = 10.5}\end{aligned}\)
Hence, the concentration of hydroxide ion can be calculated as,
\(\begin{aligned}{c\left( {O{H^ - }} \right) = {{10}^{ - pOH}}{\rm{mol}}{{\rm{L}}^{ - 1}}}\\{c\left( {O{H^ - }} \right) = {{10}^{ - 10.5}}{\rm{mol}}{{\rm{L}}^{ - 1}}}\\{c\left( {O{H^ - }} \right) = 3.2 \cdot {{10}^{ - 11}}{\rm{mol}}{{\rm{L}}^{ - 1}}}\end{aligned}\)
Therefore, the concentration of hydronium ion is \(c\left( {{H_3}{O^ + }} \right) = 3.2 \times {10^{ - 4}}mol{L^{ - 1}}\)and the concentration of hydroxide ion is\(c\left( {O{H^ - }} \right) = 3.2 \times {10^{ - 11}}{\rm{mol}}{{\rm{L}}^{ - 1}}\).


