Chapter 4: Q4.6P (page 178)
Question: Which of the following scenes best represents a volume from a solution of magnesium nitrate?
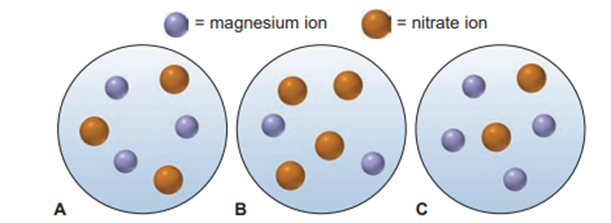
Short Answer
Answer
Scene (B) best represents a volume from a solution of magnesium nitrate.
Learning Materials
Features
Discover
Chapter 4: Q4.6P (page 178)
Question: Which of the following scenes best represents a volume from a solution of magnesium nitrate?
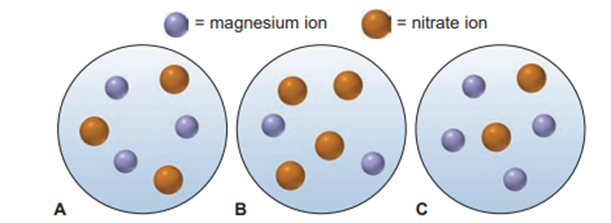
Answer
Scene (B) best represents a volume from a solution of magnesium nitrate.
All the tools & learning materials you need for study success - in one app.
Get started for free
Balance each of the following redox reactions and classify it as a combination, decomposition, or displacement reaction:
Which ions do not appear in a net ionic equation? Why?
Identify the oxidizing agent and the reducing agent in the following reaction, and explain your answer:
The brewing industry uses yeast microorganisms to convert glucose to ethanol for wine and beer. The baking industry uses the carbon dioxide produced to make bread rise:
How many grams of ethanol can be produced from 100g of glucose? What volume of CO2 is produced? (Assume 1 mol of gas occupies 22.4 L at the conditions used.)
At liftoff, a space shuttle uses a solid mixture of ammonium perchlorate and aluminum powder to obtain great thrust from the volume change of solid to gas. In the presence of a catalyst, the mixture forms solid aluminum oxide and aluminium trichloride and gaseous water and nitrogen monoxide. (a) Write a balanced equation for the reaction, and identify the reducing and oxidizing agents. (b) How many total moles of gas (water vapor and nitrogen monoxide) are produced when 50.0 kg of ammonium perchlorate reacts with a stoichiometric amount of Al? (c) What is the volume change from this reaction? (dof NH4ClO4 = 1.95 g/cc, Al = 2.70 g/cc, Al2O3 = 3.97 g/cc, and AlCl3 = 2.44 g/cc; assume 1 mol of gas occupies 22.4 L.)
What do you think about this solution?
We value your feedback to improve our textbook solutions.