Chapter 4: Q4.52P (page 180)
If 26.25 mL of a standard 0.1850 M NaOH solution is required to neutralize 25.00 mL of what is the molarity of the acid solution?
Short Answer
The molarity of the acid solution is 0.097 M.
Learning Materials
Features
Discover
Chapter 4: Q4.52P (page 180)
If 26.25 mL of a standard 0.1850 M NaOH solution is required to neutralize 25.00 mL of what is the molarity of the acid solution?
The molarity of the acid solution is 0.097 M.
All the tools & learning materials you need for study success - in one app.
Get started for free
A reaction cyclefor an element is a series of reactions beginning and ending with that element. In the following copper reaction cycle, copper has either a 0 or a _2 oxidation state. Write balanced molecular and net ionic equations for each step in the cycle.
(1) Copper metal reacts with aqueous bromine to produce a green-blue solution.
(2) Adding aqueous sodium hydroxide forms a blue precipitate.
(3) The precipitate is heated and turns black (water is released).
(4) The black solid dissolves in nitric acid to give a blue solution.
(5) Adding aqueous sodium phosphate forms a green precipitate.
(6) The precipitate forms a blue solution in sulfuric acid.
(7) Copper metal is recovered from the blue solution when zinc metal is added.
Give the oxidation number of carbon in the following:
Give the oxidation number of nitrogen in the following:
Many important compounds in the chemical industry are derivatives of ethylene . Two of them are acrylonitrile and methyl methacrylate.
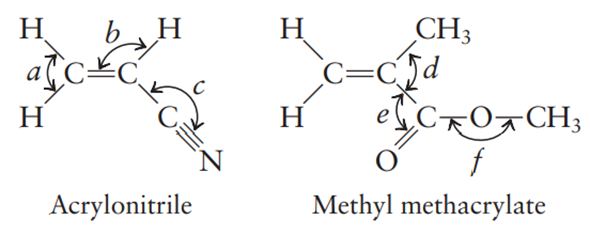
Complete the Lewis structures for these molecules, showing all lone pairs. Give approximate values for bond angles a through f, and give the hybridization of all carbon atoms. In acrylonitrile and methyl methacrylate indicate which atoms in each molecule must lie in the same plane. How many s bonds and how many p bonds are there in acrylonitrile and methyl methacrylate?
At liftoff, a space shuttle uses a solid mixture of ammonium perchlorate and aluminum powder to obtain great thrust from the volume change of solid to gas. In the presence of a catalyst, the mixture forms solid aluminum oxide and aluminium trichloride and gaseous water and nitrogen monoxide. (a) Write a balanced equation for the reaction, and identify the reducing and oxidizing agents. (b) How many total moles of gas (water vapor and nitrogen monoxide) are produced when 50.0 kg of ammonium perchlorate reacts with a stoichiometric amount of Al? (c) What is the volume change from this reaction? (dof NH4ClO4 = 1.95 g/cc, Al = 2.70 g/cc, Al2O3 = 3.97 g/cc, and AlCl3 = 2.44 g/cc; assume 1 mol of gas occupies 22.4 L.)
What do you think about this solution?
We value your feedback to improve our textbook solutions.