Chapter 4: Q4.37P (page 179)
With ions shown as spheres and solvent molecules omitted for clarity, the circle (right) illustrates the solid formed when a solution containing is mixed with one containing
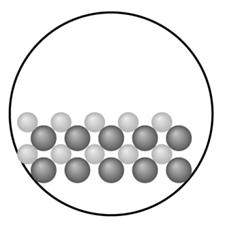
(a) Identify the solid.
(b) Write a balanced net ionic equation for the reaction.
(c) If each sphere represents of ion, what mass of product forms?
Short Answer
- The solid is
- The mass of product forms is 1.516 gm.

