Chapter 4: Q4.2P (page 140)
What two factors cause water to be polar?
Short Answer
The two factors that cause water to be polar are: the bent shape of a water molecule and its polar bonds.
Learning Materials
Features
Discover
Chapter 4: Q4.2P (page 140)
What two factors cause water to be polar?
The two factors that cause water to be polar are: the bent shape of a water molecule and its polar bonds.
All the tools & learning materials you need for study success - in one app.
Get started for free
Question:How many moles of H+ions are present in the following aqueous solutions?
(a) 1.40 L of 0.25 M perchloric acid
(b) 6.8 mL of 0.92 M nitric acid
(c) 2.6 L of 0.085 M hydrochloric acid
The allene molecule has the following Lewis structure:
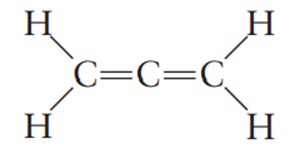
Must all four hydrogen atoms lie in the same plane? If not, what is the spatial relationship among them? Why?
In which of the following equations does sulfuric acid act as an oxidizing agent? In which does it act as an acid? Explain.
Question: Zinc hydroxide is insoluble in water but dissolves when a nitric acid solution is added. Why? Write balanced total ionic and net ionic equations, showing nitric acid as it actually exists in water and the reaction as a proton-transfer process.
The mass percent of in a seawater sample is determined by titrating 25.00 mL of seawater with solution, causing a precipitation reaction. An indicator is used to detect the endpoint, which occurs when a free ion is present in the solution after all the has reacted. If 53.63 mL of 0.2970 M is required to reach the endpoint, what is the mass percent of in the seawater (d of seawater = 1.024 g/mL)?
What do you think about this solution?
We value your feedback to improve our textbook solutions.