Chapter 4: Q4.107 P (page 182)
Describe what happens on the molecular level when acetic acid dissolves in water.
Short Answer
You need to describe what happens on the molecular level when acetic acid dissolves in water.
Learning Materials
Features
Discover
Chapter 4: Q4.107 P (page 182)
Describe what happens on the molecular level when acetic acid dissolves in water.
You need to describe what happens on the molecular level when acetic acid dissolves in water.
All the tools & learning materials you need for study success - in one app.
Get started for free
Mixtures of CaCl2 and NaCl are used to melt ice on roads. A dissolved 1.9348-g sample of such a mixture was analyzed by using excess Na2C2O4 to precipitate the Ca2+ as CaC2O4. The CaC2O4 was dissolved in sulfuric acid, and the resulting H2C2O4 was titrated with 37.68mL of 0.1019M KMnO4 solution.
(a) Write the balanced net ionic equation for the precipitation reaction.
(b) Write the balanced net ionic equation for the titration reaction. (See Sample Problem 4.11.)
(c) What is the oxidizing agent?
(d) What is the reducing agent?
(e) Calculate the mass percent of CaCl2 in the original sample.
Why must every redox reaction involve an oxidizing agent and a reducing agent?
Which of the following scenes best represents how the ions occur in an aqueous solution of:
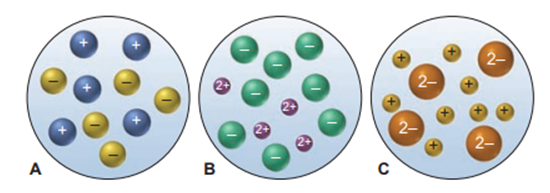
a. b. c.
In a titration of HNO3, you add a few drops of phenolphthalein indicator to 50.00 mL of acid in a flask. You quickly add 20.00 mL of 0.0502 MNaOH but overshoot the end point, and the solution turns deep pink. Instead of starting over, you add 30.00 mL of the acid, and the solution turns colorless. Then, it takes 3.22 mL of the NaOH to reach the end point. (a) What is the concentration of the HNO3solution? (b) How many moles of NaOH were in excess after the first addition?
How many total moles of ions are released when each of the following samples dissolves completely in water?(a) 0.75 mol of (b) g of (c) formula units of
What do you think about this solution?
We value your feedback to improve our textbook solutions.