Chapter 20: Q20.41 P (page 917)
Find for the formation of (l) from its elements.
Short Answer
The is obtained as: .
Learning Materials
Features
Discover
Chapter 20: Q20.41 P (page 917)
Find for the formation of (l) from its elements.
The is obtained as: .
All the tools & learning materials you need for study success - in one app.
Get started for free
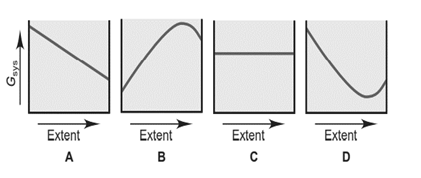
A chemical reaction, such as HI forming from its elements, can reach equilibrium at many temperatures. In contrast, a phase change, such as ice melting, is in equilibrium at a given pressure only at the melting point. Each of the graphs below depicts vs. extent of change.
(a) Which graph depicts how changes for the formation of HI? Explain.
(b) Which graph depicts how changes as ice melts at and 1atm? Explain.
Find for the reaction of nitrogen monoxide with hydrogen to form ammonia and water vapor. Is the sign ofrole="math" localid="1663358976150" as expected?
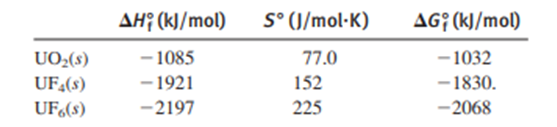
To prepare nuclear fuel, U3O8 ("yellow cake") is converted to UO2(NO3)2, which is then converted to UO3 and finally UO2.The fuel is enriched (the proportion of the is increased) by a two-step conversion of UO2 into UF6, a volatile solid, followed by a gaseous-diffusion separation of the isotopes:
Calculate for the overall process at .
a) Write a balanced equation for the gaseous reaction between N2O5 and F2 to form NF3 and O2.
b) Determine
c) Find role="math" localid="1663390048995" if
Hemoglobin carries O2 from the lungs to tissue cells, where the O2 is released. The protein is represented as Hb in its unoxygenated form and as Hb.O2 in its oxygenated form. One reason COis toxic is that it competes with O2 in binding to Hb:
(a) Ifat (body temperature), what is the ratio of [Hb.CO] to [Hb.O2] at with [O2]=[CO]?
(b) How is Le Châtelier's principle used to treat CO poisoning?
What do you think about this solution?
We value your feedback to improve our textbook solutions.