Chapter 20: Thermodynamics: Entropy, Free Energy, and the Direction of Chemical Reactions
Q20.42 P
Find for the formation of (g) from its elements.
Q20.43 P
Sulfur dioxide is released in the combustion of coal. Scrubbers use aqueous slurries of calcium hydroxide to remove the from flue gases. Write a balanced equation for this reaction and calculate role="math" localid="1663363200253" at 298 K [ of J/mol K].
Q20.44 P
Oxyacetylene welding is used to repair metal structures, including bridges, buildings, and even the Statue of Liberty. Calculate for the combustion ofmole of acetylene.
Q20.45
What is the advantage of calculating free energy changes rather than entropy changes to determine reaction spontaneity?
Q20.46 P
Given that, explain how the sign ofcorrelates with reaction spontaneity.
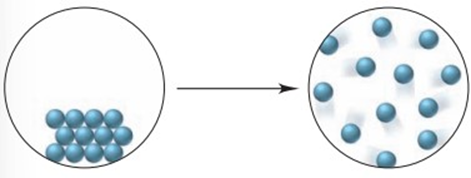
Q20.47 P
(a) Is an endothermic reaction more likely to be spontaneous at higher temperatures or lower temperatures? Explain.
(b) The change depicted below occurs at constant pressure. Explain your answers to each of the following:
(1) What is the sign of ?
(2) What is the sign of ?
(3) What is the sign of ?
(4) How does the sign ofvary with temperature?
Q20.48 P
With its components in their standard states, a certain reaction is spontaneous only at high . What do you know about the signs ofrole="math" localid="1663368103423" and? Describe a process for which this is true.
Q20.49 P
How can be relatively independent of ifof each reactant and product increases with ?
Q20.4P
State qualitatively the relationship between entropy and freedom of particle motion. Use this idea to explain why you will probably never
(a) be suffocated because all the air near you has moved to the other side of the room;
(b) see half the water in your cup of tea freeze while the other half boils.
Q20.50 P
Calculate for each reaction using values:
(a) role="math" localid="1663371231311"
(b)
(c)
