Chapter 20: Q20.48 P (page 918)
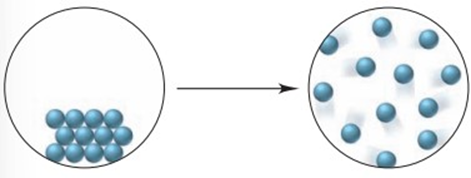
With its components in their standard states, a certain reaction is spontaneous only at high . What do you know about the signs ofrole="math" localid="1663368103423" and? Describe a process for which this is true.
Short Answer
Both, and . The entropy of the surroundings is negative: .
An example of such a process – is the evaporation of water or melting of metal.