Chapter 10: Q10.6P (page 405)
Draw a Lewis structure for
(a) ;
(b);
(c) .
Short Answer
The Lewis structure of
a.
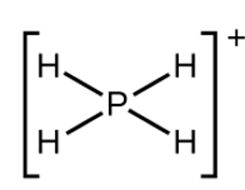
b.
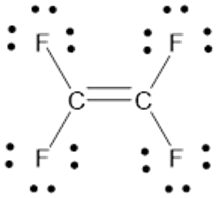
c.
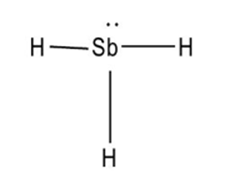
Learning Materials
Features
Discover
Chapter 10: Q10.6P (page 405)
Draw a Lewis structure for
(a) ;
(b);
(c) .
The Lewis structure of
a.

b.

c.

All the tools & learning materials you need for study success - in one app.
Get started for free
Lewis structures of mescaline, a hallucinogenic compound in peyote cactus, and dopamine, a neurotransmitter in the mammalian brain, appear below. Suggest a reason for mescaline鈥檚 ability to disrupt nerve impulses.
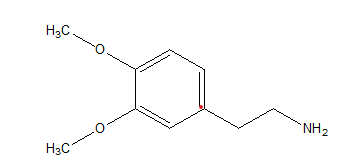
Mescaline
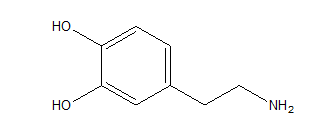
Dopamine
Dichlorine heptaoxide, , can be viewed as two groups sharing an O atom. Draw a Lewis structure for with the lowest formal charges, and predict any deviation from the ideal for the Cl-O-Cl bond angle.
Determine the electron-group arrangement, molecular shape, and ideal bond angle(s) for each of the following:
(a)
(b)
(c)
Dinitrogen difluoride, , is the only stable, simple inorganicmolecule with an N=N bond. The compound occurs incis and trans forms.(a) Draw the molecular shapes of the two forms of.(b) Predict the polarity, if any, of each form.
Which of these atoms cannot serve as a central atom in a Lewis structure?
Explain.
What do you think about this solution?
We value your feedback to improve our textbook solutions.