Chapter 10: Q10.5P (page 405)
Draw a Lewis structure for (a) ; (b) ; (c)(C is central).
Short Answer
The Lewis structure of
a.
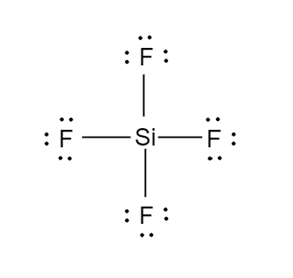
b.
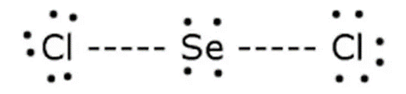
C.
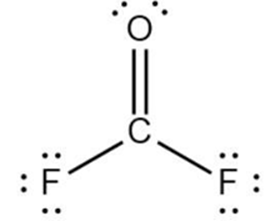
Learning Materials
Features
Discover
Chapter 10: Q10.5P (page 405)
Draw a Lewis structure for (a) ; (b) ; (c)(C is central).
The Lewis structure of
a.

b.
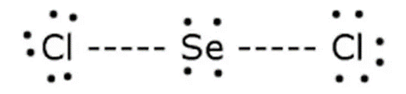
C.

All the tools & learning materials you need for study success - in one app.
Get started for free
Despite many attempts, the per-bromate ion ( ) was not prepared in the laboratory until about 1970. (In fact, articles were published explaining theoretically why it could never be prepared!) Draw a Lewis structure for which all atoms have the lowest formal charges.
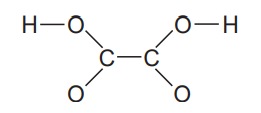
Oxalic acid ( ) is found in toxic concentrations in rhubarb leaves. The acid forms two ions, and , by the sequential loss of H ions. Draw Lewis structures for the three species, and comment on the relative lengths and strengths of their carbon-oxygen bonds. The connections among the atoms are shown below with single bonds only.
Phosgene is a colorless, highly toxic gas employed against troops in World War I and used today as a key reactant in organic syntheses. From the following resonance structures, select the one with the lowest formal charges:
Cryolite ( ) is an indispensable component in the electrochemical manufacture of aluminium. Draw a Lewis structure for the ion.
Name all the molecular shapes that have a tetrahedral electron-group arrangement.
What do you think about this solution?
We value your feedback to improve our textbook solutions.