Chapter 10: Q10.30P (page 406)
Name all the molecular shapes that have a tetrahedral electron-group arrangement.
Short Answer
The shapes that have a tetrahedral electron group arrangement are Tetrahedral, Trigonal planar, Bent shape.
Learning Materials
Features
Discover
Chapter 10: Q10.30P (page 406)
Name all the molecular shapes that have a tetrahedral electron-group arrangement.
The shapes that have a tetrahedral electron group arrangement are Tetrahedral, Trigonal planar, Bent shape.
All the tools & learning materials you need for study success - in one app.
Get started for free
Draw the Lewis structure with lowest formal charges, and determine the charge of each atom in
(a)
(b)
Arrange the following ACln species in order of decreasing Cl-A-Cl bond angles: ,data-custom-editor="chemistry" ,role="math" localid="1656921147907" , ,data-custom-editor="chemistry"
Name the shape and give the classification and ideal bond angle(s) for each of the following general molecules:
(a) 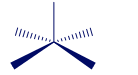
(b) 
(c)
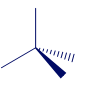
Which of the following numbers of electron groups can give rise to a bent (V-shaped) molecule: two, three, four, five, six? Draw an example for each case, showing the shape classification (AXmEn) and the ideal bond angle.
Dinitrogen monoxide ( ) supports combustion in a manner similar to oxygen, with the nitrogen atoms forming . Draw three resonance structures for (one N is central), and use formal charges to decide the relative importance of each. What correlation can you suggest between the most important structure and the observation that supports combustion?
What do you think about this solution?
We value your feedback to improve our textbook solutions.