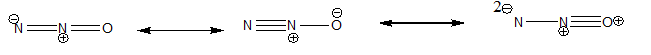Chapter 10: Q10.87 CP (page 409)
Dinitrogen monoxide ( ) supports combustion in a manner similar to oxygen, with the nitrogen atoms forming . Draw three resonance structures for (one N is central), and use formal charges to decide the relative importance of each. What correlation can you suggest between the most important structure and the observation that supports combustion?
Short Answer
The three resonance structures:

The resonance Structure (B) is the most stable structure and (C) is the least stable.
Nitrous oxide has a higher percentage of oxygen than air and easily breaks down into oxygen, which stimulates burning.