Chapter 10: Q10.3P (page 405)
In which of these bonding patterns does X obey the octet rule?
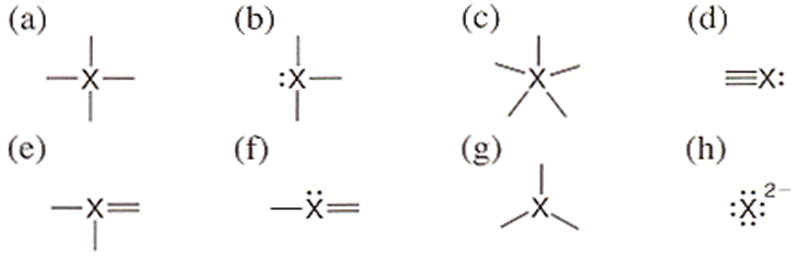
Short Answer
Expert verified
- X obeys the octet rule.
- X obeys the octet rule.
- X does not obey the octet rule.
- X obeys the octet rule.
- X obeys the octet rule.
- X obeys the octet rule.
- X does not obey the octet rule.
- X does not obey the octet rule.
Step by step solution
01
Octet Rule
The octet rule is important to attain noble gas configuration i.e., the atom should have 8 electrons in the valence shell.
It is the requirement of an electron by the molecule (by gaining an electron from another molecule or by losing its electron to another atom) or having the electron in the valence shell of the atom.
02
Subpart (a) Explanation
- The central atom X in this bonding pattern obeys the octet rule.
- It has attained the noble gas configuration as its valence shell of the central atom has 8 electrons in the valence shell after the formation of a bond with 4 different elements.
- In one bond there are two electrons. Hence, 4 bonds contain 8 electrons. Example:
03
Subpart (b) Explanation
- The central atom X in this bonding pattern obeys the octet rule.
- It has attained the noble gas configuration.
- Here, 2 electrons are present in one bond pair and 6 electrons are present in three bond pairs. Example;
04
Subpart (c) Explanation
- Here, it does not follow the noble gas configuration as it has more than 8 electrons in its valence shell.
- The existence of these type of molecule is very rare as it does not follow the basic noble gas configuration rule. Example:
05
Subpart (d) Explanation
- The central atom X in this bonding pattern obeys the octet rule.
- It has attained the noble gas configuration.
- The two atoms that form three bonds and have one lone pair that does not participate in bonding. So, the total of 8electrons in valence shell. Example:
06
Subpart (e) Explanation
- The central atom X in this bonding pattern obeys the octet rule.
- It has attained the noble gas configuration.
- The central atom forms two bonds with one atom and two single bonds with two other atoms.
- All the 8 electrons are bond pairs. Example: phosgene.
07
Subpart (f) Explanation
- The central atom X in this bonding pattern obeys the octet rule.
- It has attained the noble gas configuration.
- The central atom has 3 bond pairs and 1 lone pair. Therefore, a total of 8 electrons. Example:
08
Subpart (g) Explanation
- Here, it does not follow the noble gas configuration as it has less than 8 electrons in its valence shell.
- The compound has only 6 electrons in its outermost shell.
- The central atom has three valence electrons and forms a bond with three atoms. A total of 6 electrons are present in the valence shell. Example:
09
Subpart (h) Explanation
- Here, it does not follow the noble gas configuration as it has more than 8 electrons in its valence shell.
- The X atom contains 8 electrons already in its outermost shell and accepts two-electron more, making the total of 10 electrons in the outermost shell.
- This kind of compound exists very rarely.
- The atom has 8electrons in its valence shell and accepts two electrons more in bonding. Example;

Unlock Step-by-Step Solutions & Ace Your Exams!
-
Full Textbook Solutions
Get detailed explanations and key concepts
-
Unlimited Al creation
Al flashcards, explanations, exams and more...
-
Ads-free access
To over 500 millions flashcards
-
Money-back guarantee
We refund you if you fail your exam.
Over 30 million students worldwide already upgrade their learning with 91Ӱ��!

