Chapter 10: Q10.38 P (page 406)
Name the shape and give the classification and ideal bond angle(s) for each of the following general molecules:
(a)

(b)

(c)
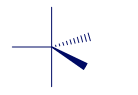
Short Answer
The shapes and bond angles of molecules can be determined from the VSEPR theory. The deviations arise mainly due to lone pair-lone pair repulsions.

