Chapter 10: The Shapes of Molecules
Q10.57P
Which molecule in each pair has the greater dipole moment?Give the reason for your choice.
Q10.58P
Which molecule in each pair has the greater dipole moment?Give the reason for your choice.
- HBr or HCl
Q10.59P
Which molecule in each pair has the greater dipole moment?Give the reason for your choice.
- HBr or HCl
Q10.5P
Draw a Lewis structure for (a) ; (b) ; (c)(C is central).
Q10.60P
Dinitrogen difluoride, , is the only stable, simple inorganicmolecule with an N=N bond. The compound occurs incis and trans forms.(a) Draw the molecular shapes of the two forms of.(b) Predict the polarity, if any, of each form.
Q10.61CP
In addition to ammonia, nitrogen forms three other hydrides: hydrazine , diazene, and tetrazene.
- Use Lewis structures to compare the strength, length, and order of nitrogen-nitrogen bonds in hydrazine, diazene, and.
- Tetrazene (atom sequence) decomposes above to hydrazine and nitrogen gas.Draw a Lewis structure for tetrazene, and calculatefor this decomposition.
Q10.62CP
Draw a Lewis structure for each species:
Q10.63CP
Give the molecular shape of each species in Problem 10.62.
Q10.64CP
Consider the following reaction of silicon tetrafluoride:
- Which depiction below best illustrates the change in molecular shape around Si?
- Give the name and designation of each shape in the depiction chosen in part (a).
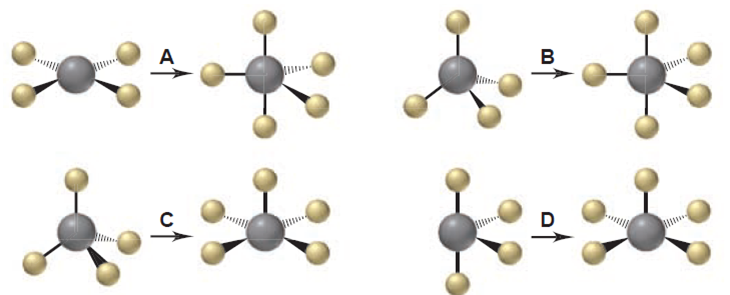
Q10.65CP
Both aluminium and iodine form chlorides, and , with “bridging” Cl atoms. The Lewis structures are
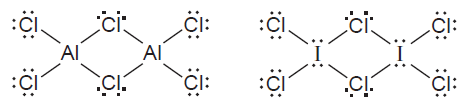
- What is the formal charge on each atom?
- Which of these molecules has a planar shape? Explain.
