Chapter 2: Q4.70P (page 40)
Give the oxidation number of manganese in the following:
Short Answer
You need to provide the oxidation no of manganese for the given compounds.
Learning Materials
Features
Discover
Chapter 2: Q4.70P (page 40)
Give the oxidation number of manganese in the following:
You need to provide the oxidation no of manganese for the given compounds.
All the tools & learning materials you need for study success - in one app.
Get started for free
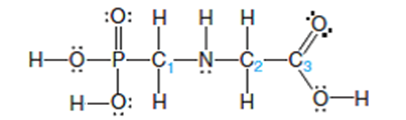
Glyphosate (below) is a common herbicide that is relatively harmless to animals but deadly to most plants. Describe the shape around and the hybridization of the P, N, and three numbered C atoms.
Use Dalton’s theory to explain why potassium nitrate from India or Italy has the same mass percents of K, N, and O.
Zinc sulfide (ZnS) occurs in the zinc blend crystal structure.How many kilograms of Zn are in 3.82 kg of ZnS?
Dolomite is a carbonate of magnesium and calcium. Analysisshows that 7.81 g of dolomite contains 1.70 g of Ca. Calculate themass percent of Ca in dolomite. On the basis of the mass percentof Ca, and neglecting all other factors, which is the richer sourceof Ca, dolomite or fluorite (see Problem 2.20)?
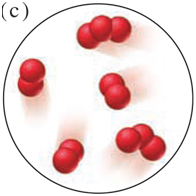
Each scene below represents a mixture. Describe each one in terms of the number of elements and/or compounds present.


What do you think about this solution?
We value your feedback to improve our textbook solutions.