Chapter 2: Q46P (page 40)
Draw atomic depictions similar to those in Problem 2.43 for
Short Answer
In an element A represents the mass number and while Z represents an atomic number of an element.
(a) (b)
(b) (c)
(c)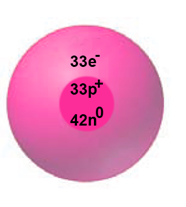
Learning Materials
Features
Discover
Chapter 2: Q46P (page 40)
Draw atomic depictions similar to those in Problem 2.43 for
In an element A represents the mass number and while Z represents an atomic number of an element.
(a) (b)
(b) (c)
(c)
All the tools & learning materials you need for study success - in one app.
Get started for free
Magnesium oxide (MgO) forms when the metal burns in the air.How many grams of Mg are in534 g of MgO?
What are some characteristic properties of elements to the left of the elements along the “staircase”? To the right?
Chlorine has two naturally occurring isotopes, 35Cl and 37Cl. What is the mass number of each isotope? How many protons, neutrons, and electrons are present in each?
A compound of iodine and cesium contains 63.94 g of metal and 61.06 g of nonmetal. How many grams of cesium are in38.77 g of compound? How many grams of iodine?
Which of Dalton’s postulates about atoms are inconsistent with later observations? Do these inconsistencies mean that Dayton was wrong? Is Dalton’s model still useful? Explain.
What do you think about this solution?
We value your feedback to improve our textbook solutions.